This Rat Tissue Inhibitors of Metalloproteinase 1 (TIMP-1) ELISA Kit is an accurate and reliable solution for measuring TIMP-1 levels in serum, plasma, cell culture supernates, and tissue homogenates of Rattus norvegicus (Rat).
This ELISA kit has been specifically designed to detect and quantify TIMP-1 levels, an important protein involved in regulating matrix metalloproteinase activity, which is a key process in cardiovascular research.Also functions as a growth factor that regulates cell differentiation, migration and cell death and activates cellular signaling cascades.
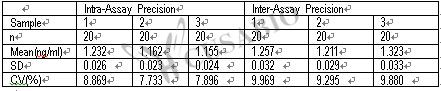
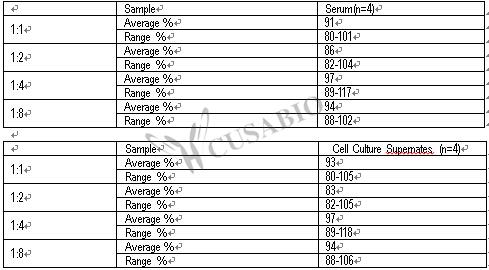
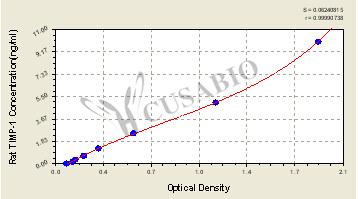
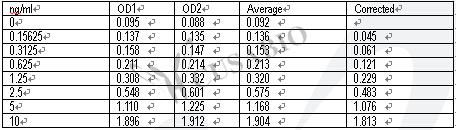
With a detection range of 0.156 ng/mL to 10 ng/mL and a sensitivity of 0.224 ng/mL, this quantitative assay can provide precise and reproducible results in just 1 to 5 hours. The sample volume required is only 50-100ul, making it an economical and efficient option for researchers.
The assay principle is based on a sandwich ELISA method, which utilizes specific antibodies to capture and detect TIMP-1 in the sample. The detection wavelength is 450 nm, ensuring accurate and reliable measurements.