In scientific research, proteins need to be added to the culture system or injected into the animal body in the form of a liquid with a certain concentration. However, many protein products are provided
lyophilized to ensure stability during shipping and on the shelf. Therefore, the lyophilized powder must be dissolved before use. We must carefully operate in the process of dissolution because poor
dissolution can lead to inactivation or inaccurate concentration of proteins, which will affect experimental results and research progress.
The purpose of this review is mainly to introduce how to properly dissolve lyophilized proteins. We emphasize the importance of lyophilization of proteins, while also, at the same time, describe in
detail the reconstituted procedures of lyophilized proteins and highlight points of concern.
2. How to Properly Dissolve Lyophilized Proteins?
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the Certificate of Analysis (CoA).
Step 1: Centrifuge the reagent tube before opening the cap. The lyophilized powder may drift and adsorb on the tube wall or the cap due to bumps during transportation.
Centrifugation helps to concentrate all the lyophilized powder at the tube bottom, which allows for completely dissolving the lyophilized powder with a small volume of liquid. Centrifugation is generally
carried out for 5 min at 3000-3500 rpm to ensure a good result.
Step 2: Dissolve the freeze-dried powder after centrifugation. This step determines whether the protein concentration is correct and whether the protein activity can reach
100%.
(1) First of all, choose the correct solution to resuspend the freeze-dried powder, generally the manual recommended solution. Some lyophilized proteins can be directly dissolved in sterile water, while
others need different buffers for resuspendsion.
The solubility of protein is related to many factors, among which pH and ionic strength are relatively important. The dissolving solution indicated in the product datasheet is the liquid that can
completely dissolve the protein. If the pH and ionic strength of the solution you use is inconsistent with that indicated in the product datasheet, in most cases, the protein can't be completely dissolved or
even can't be dissolved at all. This eventually results in partial or complete loss of activity of the prepared protein.
(2) Protein must be dissolved to the indicated concentration. Protein can maintain good stability within a certain concentration range indicated in the product datasheet, generally 0.1-1.0 mg/ ml.
However, the specific concentration range for different proteins will be distinct. Above or below this concentration range, the protein will be unstable, that is, the activity of this product will decline. It may
exceed the maximum dissolved concentration of the protein when the dissolved concentration is too high, and the protein can't be dissolved completely under this condition. Furthermore, above or below
this concentration range, protein aggregation may occur, which will still result in part of protein being undissolved and activity decline.
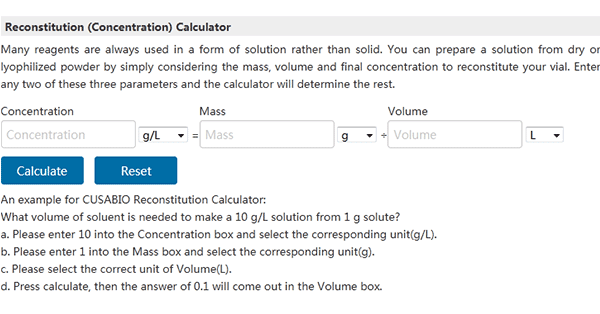
Now, you are ready to add the buffer. To ensure optimal recovery, it is recommended to reconstitute all products to the concentrations listed with a specified buffer. CUSABIO offers a helpful
reconstitution calculator at https://www.cusabio.com/m-298.html. This tool helps you easily calculate the correct volume of buffer every time.
Figure 1: How to calculate the buffer volume
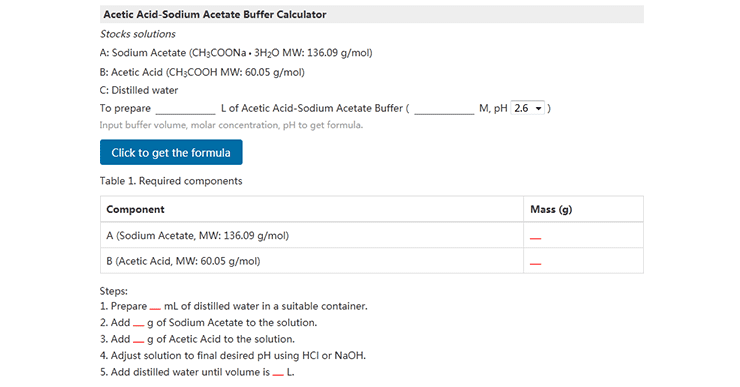
If you do not know the exact amount of buffer, CUSABIO offers a helpful buffer calculator at https://www.cusabio.com/m-296.html. It lists some
common buffers you may use for your experiments. Using the volume you calculated using the reconstitution calculator, slowly add the buffer to vial to achieve the desired concentration.
Figure 2: How to calculate the exact amount of buffer
(3) Prohibit vigorous shaking or oscillation (e.g. vortex). When mixing, avoiding shaking and vortexing, otherwise generating shear forces that result in protein loss or denaturation.
After adding the buffer, cover the bottle cap and gently blow the solution several times with the tip of the pipette gun to thoroughly dissolve the freeze-dried powder. Some insoluble protein powder
can be completely dissolved by placing them on a horizontal shaker at a low speed for a period of time. Some protein powder may be suspended for more than 2 hours at 4 ° C to achieve full dissolution.
Flakes and particulates may appear in your solution. If this happens, mix the product at room temperature for two to three hours or overnight at four degrees Celsius on a rocker platform. Place the bottle at
room temperature for at least 10 - 15 minutes before making aliquots or using them directly.
Step 3: This resuspension can be stored no more than one week at 2° C - 8 ° C.
Resuspend the protein with recommended buffer to the recommended concentration according to the manual, and then store it at 2° C - 8 ° C in the refrigerator. Under this condition, the protein
remains active for up to one week. This is sufficient for an experiment with a cycle of 5-7 days, such as the induced maturation of DC (dendritic cells). During the experiment period, just remove a certain
amount of the protein from the refrigerator and add it into the culture medium.
In fact, the recommended concentration in the manual is a little high for usual tests. So the solution should be diluted and stored at 4 ° C and then be used up within one week. For the dilution, please
operate according to the step 4 below. Use the solution containing carrier protein to conduct dilution, otherwise, the diluted cytokines or proteins will easily adhere to the tube wall or bottle wall, which will
cause the cytokines or proteins concentration to decrease and thus weaken its activity sharply.
Step 4: As for long-term storage, the proteins should be further diluted with the solution containing carrier protein (such as 0.1% BSA, 10% FBS, or 5% HAS) and then be stored at -20
° C - -80 ° C.
If the period of an experiment is longer than one week, or the prepared proteins can't be used up for one time, long-term storage is needed.
Method: Dilute the resuspended proteins with carrier protein-containing solution such as 0.1% BSA, 10% FBS, and 5% HAS, and then aliquot and store at -20°C - -80°C, that is,
conventional refrigerator freezer or ultra-low temperature refrigerator. Besides, we recommend adding 5-50% of glycerol (final concentration) and aliquot for long-term storage at -20°C - -80°C. The
resuspension should be diluted with the solution containing carrier protein before aliquot and freeze. The addition of a carrier protein, such as purified BSA (final concentration of 10-15 mg/ml), to dilute
protein solutions helps to protect against such degradation and loss. The proteins can be diluted to any concentration because the large quantity of carrier protein ensures that the protein can maintain high
stability at low concentrations.
When performing the serum-free culture or animal experiment in vivo, the proteins should contain no human or animal proteins such as BSA, FBS, or HAS. As for the long-term storage, use Trehalose
as the carrier protein, dilute the resuspended proteins with the Trehalose solution, and then aliquot and store at -20°C - -80°C. (* In medicine, trehalose has been successfully used as a stabilizer for blood products, vaccines, cells and other bioactive substances, which can not only prolong the storage time at room temperature, but also prevent blood contamination.)
In summary, the correct reconstitution of lyophilized protein powder is essential for initiating an experiment. Sometimes the results don't go well and maybe you can look in this section for reasons.



Comments
Leave a Comment