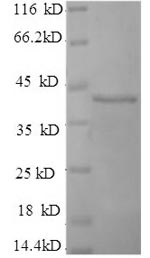
A construct encoding NH2-terminally 6xHis-tagged human selenoprotein P (SEPP1) was expressed in the E.coli cells. The resulting protein is the recombinant full-length of mature SEPP1 protein containing amino acid sequence 20-381 of mouse SEPP1. The purity of this SEPP1 protein is greater than 90% determined by SDS-PAGE. A molecular mass band range from 40 kDa was presented on the SDS-PAGE gel under reducing conditions. In-stock SEPP1 proteins are offered now. In addition to being used to immunize animals for specific antibody synthesis, this recombinant SEPP1 protein may also find use int he studies of selenium-metabolism or SEPP1-related diseases.
SEPP1 is a major selenium-containing protein in human plasma and is primarily expressed in the liver and secreted into the extracellular fluid. As a selenium-transporter, SEPP1 sustains antioxidative selenoenzymes in several tissues such as the brain and testis and plays an important role in selenium-metabolism and antioxidative defense. The reduction of SEPP1 and other selenoproteins leads to various dysfunctions associated with oxidative stress. Recent studies have shown that overexpressed SEPP1 exacerbates glucose metabolism and promotes type II diabetes.