Due to their important functions, proteins are widely used for industrial, nutritional, and medical applications. An important way to generate large amounts of a specific protein is the recombinant DNA technology, which involves the use of genetic recombination to bring together genetic material from multiple sources, creating DNA sequences that are naturally not found in the genome. Proteins produced with the recombinant DNA technology are the so-called recombinant proteins.
To help you get a better understanding of how recombinant proteins are produced, this article will cover these topics:
1. What is recombinant DNA?
To elucidate what is recombinant DNA and how to use the recombinant DNA technology to produce a specific protein, you should first know something about protein expression within the organism.
For any organism, proteins are one of the most important materials that form the body and perform essential functions. For instance, the reason why you can digest food is that your digestive tract secretes digestive enzymes, which break down macromolecules in the food into smaller molecules that could be absorbed by the body. All of these enzymes are proteins.
Since the current-structure of DNA was discovered in 1953, genetic principles have been studied extensively. Now, it's well established that proteins are made from DNA through a two-step process. The first step is called transcription, where mRNA is made from DNA. The next step is translation, where protein is made from mRNA. In other words, a DNA sequence provides instructions for making a specific protein. So gene expression actually has the same meaning as protein expression.
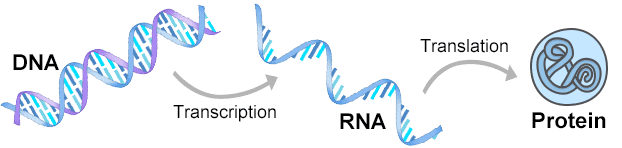
Pic. 1 Gene Expression
Recombinant DNA (rDNA) is a DNA strand that is formed by the combination of two or more DNA sequences. Genetic recombination is a naturally occurring process. The process can also be artificially manipulated for various purposes, and this is the so-called recombinant DNA technology. Using recombinant DNA technology, scientists are able to create new DNA sequences that would not naturally exist under normal circumstances and environmental conditions. The resulting recombinant DNA is put into a host cell, where it is expressed into a new protein, which is the so-called recombinant protein. The recombinant DNA technology plays a critical role in the production of recombinant proteins for pharmaceutical, medical, agricultural, and other uses.
2. How to make recombinant DNA?
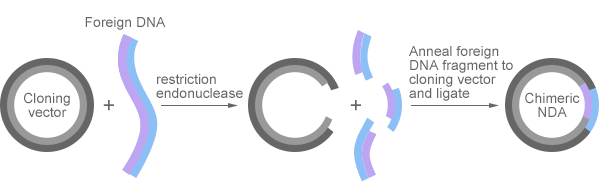
Pic. 2 Recombinant DNA
The recombinant DNA technology has other names, including DNA cloning, molecular cloning, and gene cloning. They all refer to the transfer of a foreign DNA into a self-replicating genetic element of an organism, which eventually leads to the amplification of the foreign DNA. Currently, there are three major methods to make recombinant DNA, as follow:
#1. Transformation
A foreign DNA fragment is cut and inserted into a vector, usually a plasmid. Next, the resulting vector is put into a host cell, such as the bacterium E. coli, where the foreign DNA fragment is expressed. The process of bacterial cell taking up foreign DNA is called transformation.
#2. Non-Bacterial Transformation
Non-bacterial transformation does not use bacteria as a host cell. One example is DNA microinjection, where a foreign DNA is injected directly into the nucleus of the recipient cell. Biolistics is a method in which high-velocity microprojectiles are used to help bombard foreign DNA into the recipient cell.
#3. Phage Introduction
In phage introduction, a phage is used to transfer foreign DNA into the host cell, and eventually, the phage DNA containing foreign DNA is inserted into the host cell's genome.
3. What is a recombinant protein?
Recombinant proteins are proteins that are artificially made through the recombinant DNA technology. Proteins can be used in many areas, such as diagnostic tools, vaccines, therapeutics, detergents, cosmetics, food production, and feed additives. Simply isolating proteins from their natural sources cannot meet the growing demand on proteins. The recombinant DNA technology provides a more efficient method to obtain large amounts of proteins.
This method has its advantages and disadvantages. For example, insulin, a hormone that acts as a key regulator of blood sugar and is reduced in patients with diabetes, has already been produced with the recombinant DNA technology, which saves many lives. Additionally, the recombinant DNA technology allows the manipulation of the properties of the protein of interest. In these aspects, recombinant DNA technology and recombinant proteins are beneficial. However, there are still concerns about the safety and ethics of the use of recombinant DNA technology.
4. How to produce a recombinant protein?
The production of recombinant proteins was once the domain of experts. Now, recombinant protein production has become a very mature and widespread technique, thanks to the development of simple, commercially available systems. In the process of recombinant protein production, a remaining challenge is that you will be faced with a bewildering array of choices.
Which system should be used to express the protein?
Which expression vector should be used?
Should the protein be expressed in full-length or partially?
Should the protein be tagged?
How to purify the protein?
...
You have to make a lot of decisions when producing recombinant proteins. If you choose wisely, you'll obtain high-quality recombinant proteins and the follow-up experiments are more likely to be successful. But if you make a wrong decision, you may fail to get the recombinant protein you need or the quality and purity of the recombinant protein do not meet the requirements.
Besides, since every protein is different, there is not an answer that is eternally right. How to choose among so many production strategies depends largely on the protein you intend to express.
Over the past decades, a large number of proteins from various organisms (viruses, Archaea, bacteria, and Eukarya) have been produced using the recombinant DNA technique and purified in the lab. Some researchers have summarized the several main steps in the production of recombinant proteins, as follow:
-
Obtaining the cDNA and creating the expression clone
-
Cloning
-
Expressing the protein in a suitable system
-
Small-scale test expression
-
Protein purification
-
Protein characterization
Currently, there are a variety of systems for expressing recombinant proteins, including both cell-based and cell-free systems. Cell-based systems can further classify into eukaryotic and prokaryotic systems. The five most commonly used expression strategies are as follow:
Recombinant protein expression in vitro
Recombinant protein expression in E. coli
Recombinant protein expression in yeast
Recombinant protein expression in insect-Baculovirus
Recombinant protein expression in mammalian cells
You can click these links to get more information about these systems.
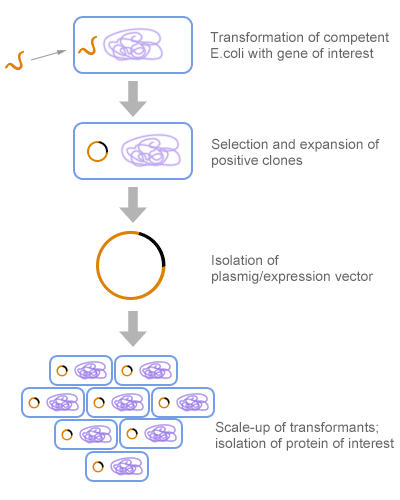
Pic. 3 Recombinant Protein Expression in E. coli
The methodology for all expression systems is fundamentally similar. The basic requirements are a DNA sequence coding for the protein of interest, a vector into which the DNA sequence is inserted, and a suitable host that will then express the foreign DNA sequence.
Despite their similar methodology, expression systems' application scope is different. Each of the five most commonly used systems has its advantages and disadvantages. So, in the production of recombinant proteins, a key consideration is the choice of expression system. Which expression system to choose depends largely on the nature of the heterologous protein to be expressed.
If you still have questions about choosing a suitable expression system, you can refer to the table 'Which expression system suits your experiment most?' on this page: https://www.cusabio.com/protein_service/
In addition to choosing a suitable expression system, how to choose a suitable expression vector is also a challenge in producing recombinant proteins. If you intend to express a protein with bioactivity, there are other things should be taken into consideration.
Many factors influence the expression of recombinant proteins. For example, we generally need a high yield of protein production, but if the recombinant protein is produced too fast, inclusion bodies may form; Many recombinant proteins need modifications like glycosylation, which are only available in eukaryotic cells, so in this case, prokaryotic cells like E. coli are not appropriate to express the proteins.
Overall, recombinant protein production is a mature technique, with various systems commercially available. But there are still challenges during the process of recombinant protein production and purification.
CUSABIO team. Production of Recombinant Protein. https://www.cusabio.com/c-20679.html






Comments
Leave a Comment