A Review of A Human NGAL ELISA Kit
Product Name:
Human neutrophil gelatinase-associated lipocalin, NGAL ELISA Kit
Code: CSB-E09408h
We used the Elisa kit CSB-E09408h for the assessment of NGAL concentration in urine samples of children, adults and chronic kidney disease patient communities. The detection range of the kit was 15.6 pg/mL-1000 pg/mL and based on preliminary studies we identified the NGAL levels of these groups are higher than the upper detection limit of the kit. Hence, we adopted sample dilution for NGAL assay.
We used 4 fold and 5 fold dilutions of samples for the analysis. Following dilution, the detection limit of the kit is adequate for the assessment of NGAL in healthy subjects. However, in patients with severe kidney injury and chronic kidney disease (CKD) / CKD of uncertain etiology (CKDu) are known to express, NGAL at elevated levels, particularly in the later stages. In such cases sample dilution at a higher ratio may be required before analysis. We analyzed urine samples of nearly 1000 children in duplicate using the Cusabio ELISA kits.
According to our assays, the intra-assay precision ranged from 6.1 % to 8.8 % while the inter assay precision ranged from 6.8 % to 8.7 % (Figure 1).
The experimental inter assay and intra assay precision were within the specified range by the manufacturer. The ELISA kit and assay protocol is designed in a very convenient manner. In the final phase, color development was achieved within 10-15 minutes. Absorbance of the blank was less than 0.3 and absorbance at the highest concentration of the standard series was around 3.6. For absorbance measurements the main wavelength was 450 nm and we used a reference wavelength of 540 nm to enhance accuracy of the measurements.
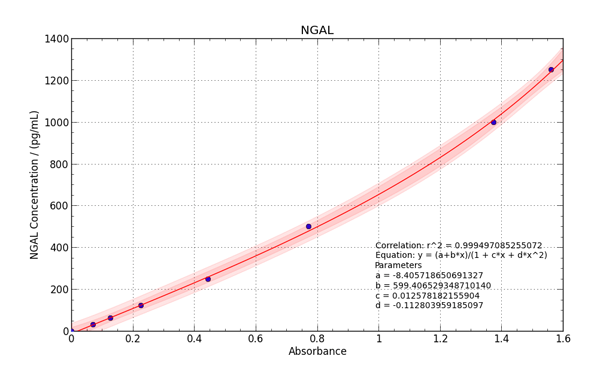
Figure 1. Human NGAL standard curve in urine sample
We used 4 four parameter logistic regression model for the construction of the standard curve using the software recommended by the manufacturer. We received precise standard curves with correlation coefficient (r) higher than 0.99 in each assay (Figure 1).
In further analysis recovery for the standard curve ranged between 92 % to 109 %, indicating a greater degree of accuracy of standard curve. We added samples and standards to the assay plate in duplicate. As we observed, there was no significant difference between absorbance values of the duplicates. Thus, we were able to determine biomarker concentrations in samples with a greater degree of accuracy with Cusabio ELISA kits.
As we identified Cusabio ELISA kits to produce great accuracy and precision in results, particularly we used Cusabio ELISA kits also for determination of pediatric reference intervals for urinary NGAL, which requires a greater degree of reliability.
The ELISA kit contained two vials containing standards and diluents in quite adequate amounts. The contents were well packed with a detailed instruction manual that can be easily understood. Following payment confirmation, Cusabio arranged the shipment within a few days and ensured fastest possible delivery of the kits. The kits were well packed with high thermal insulation with frozen gel packs to ensure thermal protection of the kits during shipment. Customer service was also very friendly and responsive. We are very satisfied about the quality of the products and customer support, and we highly recommend Cusabio ELISA kits for researchers.
By Sameera Gunasekara
University of Ruhuna, Sri Lanka
Cite this article
CUSABIO team. A Review of A Human NGAL ELISA Kit. https://www.cusabio.com/c-21084.html




Comments
Leave a Comment