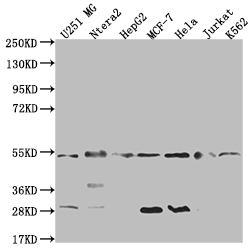
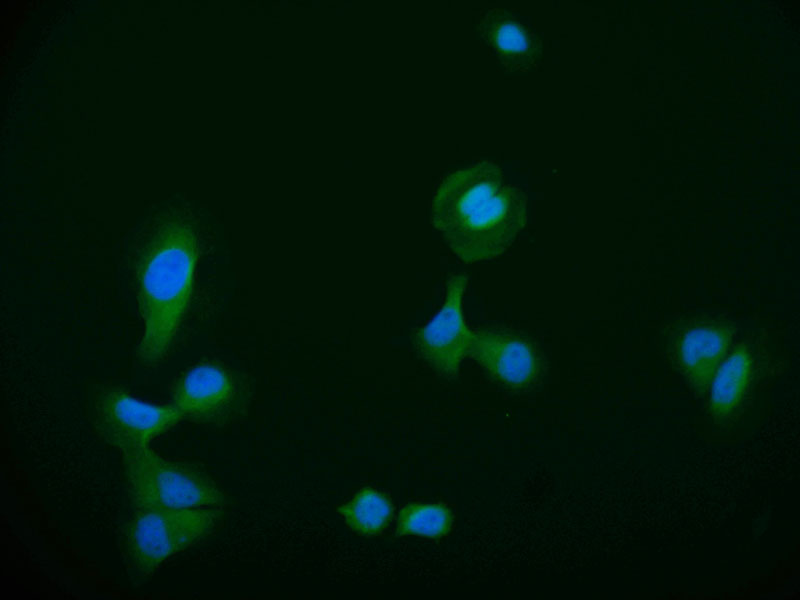
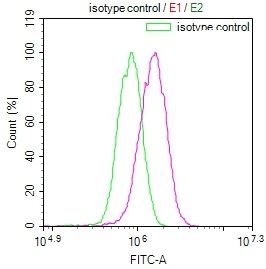
The production of the DNAJA1 recombinant monoclonal antibody follows a meticulous and standardized process to ensure its quality and specificity. B cells are initially isolated from an immunized animal, with the synthesized peptide derived from human DNAJA1 used as the immunogen. Total RNA is then extracted from the isolated B cells and converted into cDNA through reverse transcription. The DNAJA1 antibody genes are amplified using PCR with specific primers designed for the antibody constant regions and subsequently inserted into an expression vector. The expression vector is introduced into host cells, allowing for the production of the DNAJA1 recombinant monoclonal antibody. The antibody is harvested from the cell culture supernatant and purified using affinity chromatography, resulting in a highly purified form. Extensive characterization assays, including ELISA, WB, IF, and FC analysis, are performed to validate the antibody's specificity and functionality, ensuring its precise binding to human DNAJA1 protein.