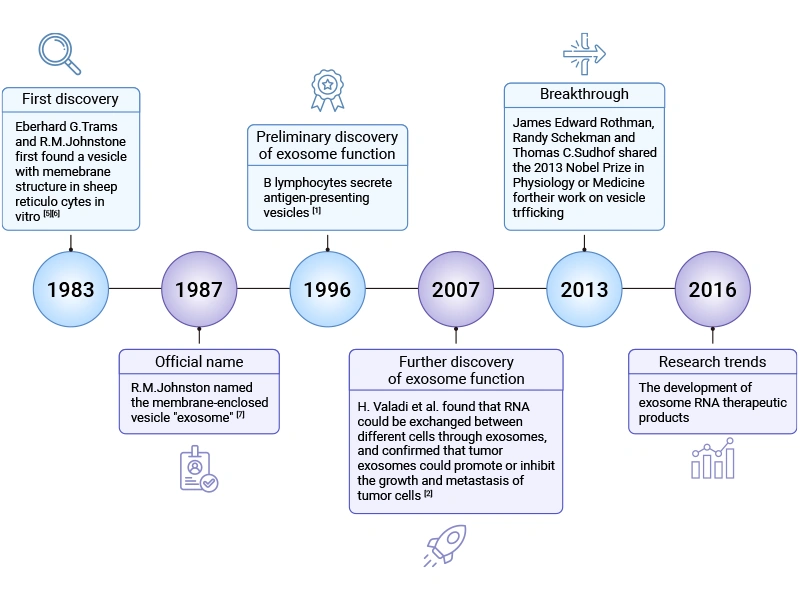
[1] G. Raposo, H.W. Nijman, W. Stoorvogel, R. Liejendekker, C.V. Harding, C.J. Melief, H.J. Geuze. B lymphocytes secrete antigen-presenting vesicles [J]. J. Exp. Med. 1996, 183:1161–1172.
[2] H. Valadi, A. Bossios, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells [J]. Nat. Cell Biol. 2007, 9:654–659.
[3] Zong SF, Wang L, et al. Facile detection of tumor-derived exosomes using magnetic nanobeads and SERS nanoprobes [J]. Anal Methods. 2016, 8:5001–08.
[4] Harding CV, Heuser JE, et al. Exosomes: looking back three decades and into the future [J]. J. Cell Biol. 2013, 200:367–371.
[5] B.T. Pan, R.M. Johnstone. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor [J]. Cell. 1983, 33:967–978.
[6] Pan BT, Teng K, et al. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes [J]. J. Cell Biol.1985;101:942–948.
[7] Johnstone RM, Bianchini A, et al. Reticulocyte maturation and exosome release: transferrin receptor containing exosomes shows multiple plasma membrane functions [J]. Blood. 1989, 74:1844–1851.
[8] Li, Q., Ding, Y., et al. (2026). 80 years of extracellular vesicles: From discovery to clinical translation [J]. Extracellular Vesicles and Circulating Nucleic Acids, 7(1), 165.
[9] Omrani, M., Beyrampour-Basmenj, H., et al. (2023). Global trend in exosome isolation and application: An update concept in management of diseases [J]. Molecular and Cellular Biochemistry, 1.
[10] Li, M., Rai, A. J., et al. (2015). An optimized procedure for exosome isolation and analysis using serum samples: Application to cancer biomarker discovery [J]. Methods, 87, 26-30.
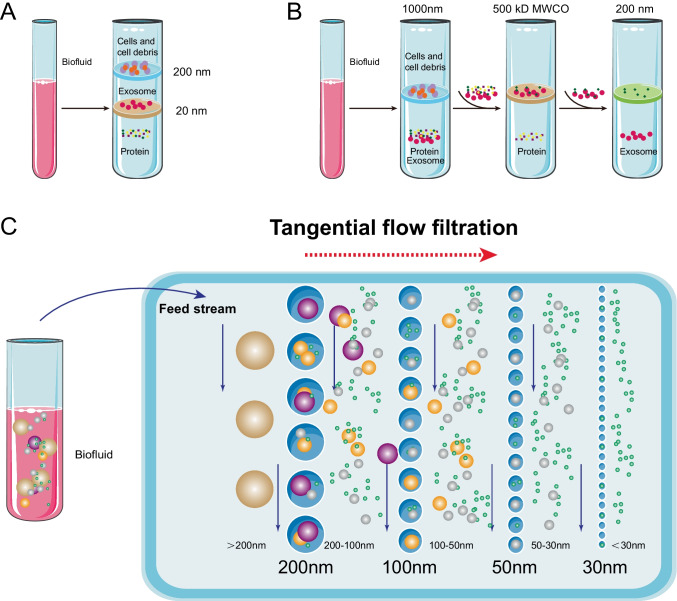
[11] Contreras-Naranjo, J. C., Wu, H. J., & Ugaz, V. M. (2017). Microfluidics for exosome isolation and analysis: Enabling liquid biopsy for personalized medicine [J]. Lab on a Chip, 17(21), 3558.
[12] Jiawei, S., Zhi, C., Kewei, T., & Xiaoping, L. (2022). Magnetic bead-based adsorption strategy for exosome isolation [J]. Frontiers in Bioengineering and Biotechnology, 10, 942077.
[13] Xu, K., Jin, Y., Li, Y., Huang, Y., & Zhao, R. (2022). Recent Progress of Exosome Isolation and Peptide Recognition-Guided Strategies for Exosome Research [J]. Frontiers in Chemistry, 10, 844124.
[14] Liu, W. Z., Ma, Z. J., & Kang, X. W. (2022). Current status and outlook of advances in exosome isolation [J]. Analytical and Bioanalytical Chemistry, 414(24), 7123.
[15] Dilsiz, N. (2024). A comprehensive review on recent advances in exosome isolation and characterization: Toward clinical applications [J]. Translational Oncology, 50, 102121.
[16] Wang, J., Ma, P., Kim, D. H., Liu, B. F., & Demirci, U. (2021). Towards Microfluidic-Based Exosome Isolation and Detection for Tumor Therapy [J]. Nano Today, 37, 101066.
[17] Cheng, Y., Qu, X., et al. (2020). Comparison of serum exosome isolation methods on co-precipitated free microRNAs [J]. PeerJ, 8, e9434.
[18] Gao M, Cai J, Zitkovsky HS, Chen B, Guo L. Comparison of Yield, Purity, and Functional Properties of Large-Volume Exosome Isolation Using Ultrafiltration and Polymer-Based Precipitation. Plast Reconstr Surg. 2022 Mar 1;149(3):638-649.
[19] Pölzl L, Nägele F, et al. Exosome Isolation after in vitro Shock Wave Therapy [J]. J Vis Exp. 2020 Sep 10;(163).
[20] Abramowicz, A., Marczak, L., et al. (2018). Harmonization of exosome isolation from culture supernatants for optimized proteomics analysis [J]. PLoS ONE, 13(10), e0205496.
[21] Neerukonda, S. N., Egan, N. A., et al. (2020). A comparison of exosome purification methods using serum of Marek's disease virus (MDV)-vaccinated and -tumor-bearing chickens [J]. Heliyon, 6(12), e05669.
[22] Tangwattanachuleeporn, M., Muanwien, P., Teethaisong, Y., & Somparn, P. (2022). Optimizing Concentration of Polyethelene Glycol for Exosome Isolation from Plasma for Downstream Application [J]. Medicina, 58(11), 1600.
[23] Lobb, R. J., Becker, M., et al. (2015). Optimized exosome isolation protocol for cell culture supernatant and human plasma [J]. Journal of Extracellular Vesicles, 4, 10.3402/jev.v4.27031.
[24] Doyle, L. M., & Wang, M. Z. (2019). Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis [J]. Cells, 8(7), 727.
[25] Zhang, H., & Lyden, D. (2019). Asymmetric flow field-flow fractionation technology for exomere and small extracellular vesicle separation and characterization [J]. Nature Protocols, 14(4), 1027.
[26] Zhang H, Freitas D, Kim HS, et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation [J]. Nat Cell Biol. 2018;20(3):332–343.
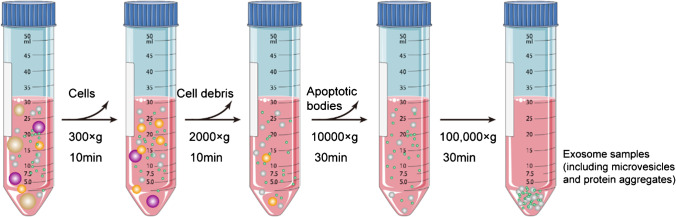
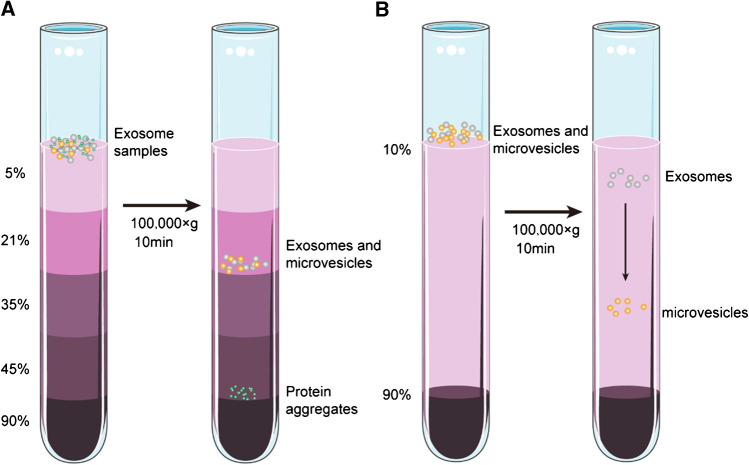
[27] Greening DW, Xu R, Ji H, Tauro BJ, Simpson RJ. A protocol for exosome isolation and characterization: evaluation of ultracentrifugation density-gradient separation, and immunoaffinity capture methods [J]. Methods Mol Biol. 2015;1295:179–209.
[28] Iorember FM, Vehaskari VM. Uromodulin: old friend with new roles in health and disease [J]. Pediatr Nephrol. 2014;29(7):1151–1158.
[29] Liu F, Vermesh O, Mani V, Ge TJ, et al. The exosome total isolation chip [J]. ACS Nano. 2017;11(11):10712–10723.
[30] Yang Q, Cheng L, Hu L, et al. An integrative microfluidic device for isolation and ultrasensitive detection of lung cancer-specific exosomes from patient urine [J]. Biosens Bioelectron. 2020;163:112290.
[31] Santana SM, Antonyak MA, Cerione RA, Kirby BJ. Microfluidic isolation of cancer-cell-derived microvesicles from hetergeneous extracellular shed vesicle populations [J]. Biomed Microdevices. 2014;16(6):869–877.
[32] Wunsch BH, Smith JT, et al. Nanoscale lateral displacement arrays for the separation of exosomes and colloids down to 20 nm [J]. Nat Nanotechnol. 2016;11(11):936–940.
[33] Ibsen SD, Wright J, et al. Rapid isolation and detection of exosomes and associated biomarkers from plasma [J]. ACS Nano. 2017;11(7):6641–6651.
[34] Zhao W, Zhang L, et al. Microsphere mediated exosome isolation and ultra-sensitive detection on a dielectrophoresis integrated microfluidic device [J]. Analyst. 2021;146(19):5962–5972.
[35] Wu M, Ouyang Y, Wang Z, et al. Isolation of exosomes from whole blood by integrating acoustics and microfluidics [J]. Proc Natl Acad Sci U S A. 2017;114(40):10584–10589.
[36] Wang Z, Li F, Rufo J, et al. Acoustofluidic salivary exosome isolation: a liquid biopsy compatible approach for human papillomavirus-associated oropharyngeal cancer detection [J]. J Mol Diagn. 2020;22(1):50–59.
[37] Pang B, Zhu Y, Ni J, et al. Extracellular vesicles: the next generation of biomarkers for liquid biopsy-based prostate cancer diagnosis [J]. Theranostics. 2020;10(5):2309–2326.
[38] Wang Z, Wu HJ, et al. Ciliated micropillars for the microfluidic-based isolation of nanoscale lipid vesicles [J]. Lab Chip. 2013;13(15):2879–2882.
[39] Ranjan S, Zeming KK, Jureen R, Fisher D, Zhang Y. DLD pillar shape design for efficient separation of spherical and non-spherical bioparticles [J]. Lab Chip. 2014;14(21):4250–4262.
[40] Zhu H, Lin X, Su Y, Dong H, Wu J. Screen-printed microfluidic dielectrophoresis chip for cell separation [J]. Biosens Bioelectron. 2015;63:371–378.
[41] Aghilinejad A, Aghaamoo M, Chen X, Xu J. Effects of electrothermal vortices on insulator-based dielectrophoresis for circulating tumor cell separation [J]. Electrophoresis. 2018;39(5–6):869–877.
[42] Fraunhofer W, Winter G. The use of asymmetrical flow field-flow fractionation in pharmaceutics and biopharmaceutics [J]. Eur J Pharm Biopharm. 2004;58(2):369–383.
[43] Wu B, Chen X, Wang J, et al. Separation and characterization of extracellular vesicles from human plasma by asymmetrical flow field-flow fractionation [J]. Anal Chim Acta. 2020;1127:234–245.
[44] Kim YB, Yang JS, Lee GB, Moon MH. Evaluation of exosome separation from human serum by frit-inlet asymmetrical flow field-flow fractionation and multiangle light scattering [J]. Anal Chim Acta. 2020;1124:137–145.
[45] Witwer, K. W., Goberdhan, D. C., et al. (2021). Updating MISEV: Evolving the minimal requirements for studies of extracellular vesicles [J]. Journal of Extracellular Vesicles, 10(14), e12182.
[46] Patel GK, Khan MA, et al. Comparative analysis of exosome isolation methods using culture supernatant for optimum yield, purity and downstream applications [J]. Sci Rep. 2019 Mar 29;9(1):5335.
[47] Aliakbari F, Stocek NB, et al. A methodological primer of extracellular vesicles isolation and characterization via different techniques [J]. Biol Methods Protoc. 2024 Feb 13;9(1):bpae009.
[48] Pang B, Zhu Y, et al. Quality Assessment and Comparison of Plasma-Derived Extracellular Vesicles Separated by Three Commercial Kits for Prostate Cancer Diagnosis [J]. Int J Nanomedicine. 2020 Dec 15;15:10241-10256.

![Typical representative composition of exosomal cargo [14]](https://www.cusabio.com/manage/upload/202604/Exosomal-cargoes.webp)


Comments
Leave a Comment