Streptococcus pneumoniae (S. pneumoniae) is a major cause of pneumonia, meningitis, sepsis and other serious illnesses worldwide. In November 2010, the WHO announced that between 700,000 and 1 million children under the age of five die of pneumococcal disease each year worldwide, and in 2015 that number will drop to 300,000. But even so, seven children under the age of five suffer from pneumococcal disease caused by streptococcus pneumoniae every minute. So what is S. pneumoniae? And who is at risk? Continuing to read this article…
1. What is Streptococcus Pneumoniae?
S. pneumoniae, also known as pneumococcus, are gram-positive, facultative anaerobic member of the genus Streptococcus [1]. In 1881, G. Stermberg in the U.S. and L. Pasteur in France isolated the bacterium, which was originally named Pneumococcus. It was officially named Streptococcus pneumoniaein 1974 because of its close resemblance to Streptococcus. S. pneumoniae is usually shaped like slightly pointed cocci. Individual S. pneumoniae bacterium usually measures between 0.5 and 1.25 micrometers in diameter. Typically, Streptococcus Pneumoniae is completely enclosed by polysaccharide capsules, which makes it such an effective virus. Its cell wall is made up of peptidoglycan, about six layers thick, and lipoteichoic acid which is attached to the membrane by a lipid moiety.
The genome of S. pneumoniae is a closed, circular DNA structure that contains between 2.0 and 2.1 million base pairs depending on the strain. It has a core set of 1553 genes, plus 154 genes in its virulome, which contribute to virulence and 176 genes that maintain a noninvasive phenotype. Among different types of strains, genetic information can vary up to 10% [2].
2. What are The Diseases Caused by Streptococcus Pneumoniae Infection?
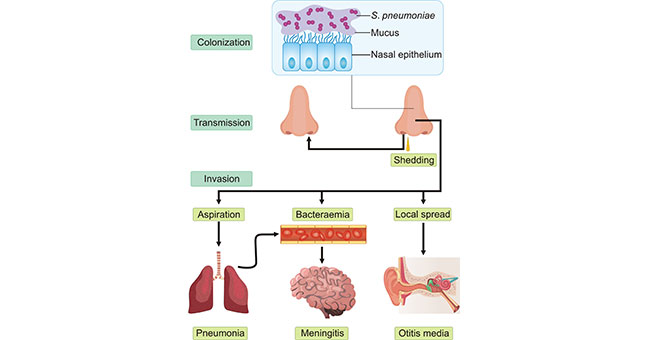
S. pneumoniae is extracellular, opportunistic pathogen that colonizes the mucosal surfaces of the human upper respiratory tract (URT), and may be isolated from the nasopharynx of 5–90% of healthy persons, depending on the population and setting. Up to 27–65% of children and <10% of adults are carriers of S. pneumoniae and carriage involves a commensal relationship between the bacterium and the host. Local spread, aspiration or seeding to the bloodstream results in invasive inflammatory diseases [3] [4] [5] (Fig. 1). S. pneumoniae is a leading bacterial cause of a wide range of infections, including otitis media, pneumonia, sepsis and meningitis. Among of them, pneumonia is the most common disease caused by S. pneumoniae infection. Additionally, it can also cause invasive Streptococcus pneumoniae disease in sterile parts of the central nervous system, abdominal cavity, joints, heart valves, and pericardium through blood circulation. As all of these diseases are 'dead ends' in the life cycle of the organism, the bacterial factors that cause invasive diseases must also be adaptive for colonization and transmission.
Figure 1. The life cycle of Streptococcus pneumoniae and the pathogenesis of pneumococcal disease
*This figure is derived from the publication on Nat. Rev. Microbiol. [6]
Regarding of pneumonia caused by S. pneumoniae infection, it is more common during winter and early spring. In tropical climates with dry and rainy seasons, pneumococcal disease tends to occur more in the dry season. Moreover, travelers are more likely to get pneumococcal disease if they spend time in crowded settings or in close contact with children in countries where pneumococcal vaccine is not routinely used.
3. Pathogenic Mechanisms of Streptococcus pneumoniae
Streptococcus pneumoniae, a Gram-positive coccus, employs a multifaceted approach in its pathogenic mechanisms, involving several intricate steps, including adhesion, invasion, resistance to phagocytosis, release of virulence factors, and the establishment of a pathogenic phenotype.
Adhesion: The pathogenicity of S. pneumoniae initiates with its ability to adhere to the surface of host cells. This is mediated by various proteins and polysaccharide structures on the bacterial surface. Notably, the capsule of S. pneumoniae is a crucial factor, as it can inhibit adhesion and aid the bacteria in evading the host's immune attacks.
Invasion: S. pneumoniae invades host cells and tissues through multiple mechanisms, with a significant one being the invasion of upper respiratory epithelial cells. Bacteria utilize surface proteins like PspC to interact with host cells, facilitating their entry into the cells.
Phagocytosis Resistance: S. pneumoniae possesses the capability to resist attacks from host phagocytic cells, achieved through various mechanisms. For instance, it can interfere with the phagocytosis process of neutrophils and macrophages, enabling the bacteria to escape engulfment.
Release of Virulence Factors: S. pneumoniae produces various virulence factors, including hemolysins, proteases, and its autolysin. These factors contribute to the bacteria escaping the phagosomal compartment, penetrating tissues, and inducing an inflammatory response at the site of infection.
Pathogenic Phenotype: The pathogenic phenotype of S. pneumoniae is closely linked to its ability to withstand attacks from the host immune system. The capsule, again, emerges as a pivotal pathogenic factor, aiding in immune evasion and enhancing the bacterium's survival within the host.
Understanding molecular-level information about the genome, metabolic pathways, and gene expression regulation of S. pneumoniae is crucial for a thorough investigation of its biological characteristics. Such research contributes to unveiling its pathogenic mechanisms and facilitates the development of novel drugs and vaccines.
4. Research on Anti-Infective Therapies
Investigating the impact of Streptococcus pneumoniae-induced infections on the host immune system and exploring new anti-infective treatment methods is a critical research direction in the field of Streptococcus pneumoniae. This includes:
4.1 Mechanisms Affecting the Host Immune System
a. Inflammatory Response: Infection by Streptococcus pneumoniae triggers an inflammatory response in the host immune system, involving inflammatory cell infiltration and the release of cytokines mediating inflammation. While this process is a normal physiological response for the host to combat infection, Streptococcus pneumoniae can employ mechanisms to evade immune system surveillance and attacks.
b. Phagocytosis Resistance: Streptococcus pneumoniae can evade the immune system's clearance by inhibiting the phagocytic activity of neutrophils and macrophages. This involves the action of surface factors such as extracellular polysaccharides and surface proteins, making bacteria more resistant to engulfment and degradation.
c. Immune Evasion: The capsule of Streptococcus pneumoniae is a crucial factor in immune evasion, inhibiting the phagocytic process of macrophages and slowing down immune system recognition. Capsular variation enables Streptococcus pneumoniae to avoid attacks from the immune system, facilitating the establishment of infection within the host.
4.2 Exploring New Anti-Infective Treatment Methods
a. Vaccine Development: Vaccination is an effective means of preventing infection. Researchers are dedicated to developing more effective vaccines against Streptococcus pneumoniae, including vaccines targeting multiple serotypes to enhance the vaccine's broad-spectrum coverage and immune protection.
b. Novel Antibiotics: Given the increasing antibiotic resistance, finding new antibiotics is a focal point of current research. Some studies explore new antibiotic targets, such as novel metabolic pathways of Streptococcus pneumoniae or bacterial tolerance mechanisms, to address the limitations of traditional antibiotics.
c. Immunomodulatory Therapy: Researchers are also considering treating infections by modulating the host immune system. This may involve enhancing the host's immune response for more effective bacterial clearance or regulating excessive inflammatory responses to alleviate infection-induced damage.
d. Combination Drug Therapy: Some studies explore combinations of different drugs to enhance treatment efficacy and slow down the development of resistance. This may involve simultaneous interventions targeting multiple pathways to increase antibacterial effectiveness.
5. Recent Research and Advances
(Zhang et al., 2019) analyzed mNGS for Streptococcus pneumoniae (S. pneumoniae) identification in paediatric bacterial meningitis, revealing a 25.9% positive rate using Alere BinaxNow®Streptococcus pneumoniae Antigen test in 135 cases [7].
(Sharew et al., 2021) aimed to assess antimicrobial resistance in S. pneumoniae isolates from pneumococcal infection-suspected patients in Ethiopia. The study confirmed Streptococcus pneumoniae through various tests and highlighted the global challenge of antimicrobial-resistant strains due to inappropriate antibiotic use [8].
(McNeil et al., 2021) evaluated the indirect impact of COVID-19 prevention measures on invasive Staphylococcus aureus, Streptococcus pneumoniae, and Group A Streptococcus in Houston area children. The study observed a decline in invasive pneumococcal disease and Group A Streptococcus associated with social distancing/masking/school closures [9].
(Shearer et al., 2021) investigated the role of the thiol glutathione in HOSCN resistance in Streptococcus pneumoniae, the leading cause of community-acquired pneumonia globally [10].
(Tian et al., 2020) explored the effects of neonatal S. pneumoniae pneumonia on vitamin A expressions and the impact of vitamin A supplementation after neonatal pneumonia on adulthood asthma development [11].
CUSABIO Recombiant Proteins of Streptococcus pneumoniae for research
References
[1] Ryan KJ, Ray CG, eds. Sherris Medical Microbiology. McGraw Hill. 2004, ISBN 978-0-8385-8529-0.
[2] van der Poll T, Opal SM. Pathogenesis, treatment, and prevention of pneumococcal pneumonia [J]. Lancet. 2009, 374 (9700): 1543–56.
[3] Abdullahi, O. et al. The prevalence and risk factors for pneumococcal colonization of the nasopharynx among children in Kilifi District, Kenya [J]. PLoS ONE. 2012, 7, e30787.
[4] Yahiaoui, R. Y. et al. Prevalence and antibiotic resistance of commensal Streptococcus pneumoniae in nine European countries [J]. Future Microbiol. 2016, 11, 737–744.
[5] Bogaert, D., De Groot, R. & Hermans, P. W. Streptococcus pneumoniae colonisation: the key to pneumococcal disease. Lancet Infect. Dis. 2004, 4, 144–154.
[6] Jeffrey N. Weiser, Daniela M. Ferreira and James C. Paton. Streptococcus pneumoniae: transmission, colonization and invasion [J]. Nat. Rev. Microbiol. 2018.
[7] Xi-Xi Zhang; Ling-Yun Guo; Lin-Lin Liu; Ao Shen; Wen-Ya Feng; Wen-Hua Huang; Hui-Li Hu; Bing Hu; Xin Guo; Tian-Ming Chen; He-Ying Chen; Yong-Qiang Jiang; Gang Liu; "The Diagnostic Value of Metagenomic Next-generation Sequencing for Identifying Streptococcus Pneumoniae in Paediatric Bacterial Meningitis", BMC INFECTIOUS DISEASES, 2019. (IF: 3)
[8] Bekele Sharew; Feleke Moges; Gizachew Yismaw; Wondwossen Abebe; Surafal Fentaw; Didrik Vestrheim; Belay Tessema; "Antimicrobial Resistance Profile and Multidrug Resistance Patterns of Streptococcus Pneumoniae Isolates from Patients Suspected of Pneumococcal Infections in Ethiopia", ANNALS OF CLINICAL MICROBIOLOGY AND ANTIMICROBIALS, 2021. (IF: 3)
[9] J Chase McNeil; Anthony R Flores; Sheldon L Kaplan; Kristina G Hulten; "The Indirect Impact of The SARS-CoV-2 Pandemic on Invasive Group A Streptococcus, Streptococcus Pneumoniae and Staphylococcus Aureus Infections in Houston Area Children", THE PEDIATRIC INFECTIOUS DISEASE JOURNAL, 2021. (IF: 3)
[10] Heather L Shearer; James C Paton; Mark B Hampton; Nina Dickerhof; "Glutathione Utilization Protects Streptococcus Pneumoniae Against Lactoperoxidase-derived Hypothiocyanous Acid", FREE RADICAL BIOLOGY & MEDICINE, 2021. (IF: 3)
[11] Yonglu Tian; Qinqin Tian; Yi Wu; Xin Peng; Yunxiu Chen; Qinyuan Li; Guangli Zhang; Xiaoyin Tian; Luo Ren; Zhengxiu Luo; "Vitamin A Supplement After Neonatal Streptococcus Pneumoniae Pneumonia Inhibits The Progression of Experimental Asthma By Altering CD4+T Cell Subsets", SCIENTIFIC REPORTS, 2020. (IF: 3)
CUSABIO team. Streptococcus pneumoniae, a Primary Cause of Pneumonia. https://www.cusabio.com/c-20998.html



Comments
Leave a Comment