Product Name:
WFDC8 antibody
Code: CSB-PA818226LA01HU
I am Dr. Ferran Barrachina, currently a postdoctoral fellow at the Massachusetts General Hospital and Harvard Medical School in Boston (United States). My research background is in reproductive biology and immunology.
For this project, we were particularly focused on deciphering whether extracellular vesicles from the epididymis, named epididymosomes, were implicated in the protein transfer from the epididymis to spermatozoa. By performing an integrative analysis using both human and mouse data we identified sperm proteins with a potential epididymis-derived origin, and the protein WFDC8 was one of the selected candidates to be validated. We found online that CUSABIO had an antibody against WFDC8 and with species reactivity in humans. Derived from our results, we demonstrated through immunofluorescence that this antibody against WFDC8 works well for human and mice.
Immunofluorescence using the WFDC8 antibody was performed in both human and mouse epididymis and testis tissues, and in mouse sperm and extracellular vesicles isolated from the epididymis (epididymosomes). For the human paraffin-embedded sections, the process included a xylene deparaffinization (2 x 100% xylene), followed by washes in ethanol (2 x 100% EtOH), and tissue rehydration through graded series of ethanol (90%, 70%, 50%, 30%). Afterward, all samples (including human/mouse testes and epididymides, and mouse sperm alone or incubated with epididymosomes) were hydrated in PBS and, for antigen retrieval, samples were treated with PBS containing 1% SDS and 0.1% Triton for 4min. The anti-WFDC8 antibody required an additional antigen retrieval step, and slides were microwaved for 1 min in 1mM EDTA, 10mM Tris (pH 9.0). Slides were blocked in 1% bovine serum albumin, and incubated with the primary antibody WFDC8 (protein G purified rabbit polyclonal antibody against the WAP four-disulfide core domain protein 8; WFDC8; 0.08 mg/ml; #CSB-PA818226LA01HU, Cusabio Technology LLC, Houston, TX, USA) at 4C for 16 h. The following secondary antibodies from Jackson Immunoresearch Laboratories (West Grove, PA, USA) were used: goat anti-rabbit IgG conjugated to Alexa Fluor 488 (30 ug/ml; #111-545-144) and donkey anti-rabbit IgG conjugated to Cy3 (7.5 ug/ml; #711-165-152). All antibodies were diluted in DAKO medium (Dako, Carpinteria, CA, USA). For the negative controls, the incubations were performed with the secondary antibody only. Slides were mounted using SlowFadeTM Diamond Antifade Mountant (Invitrogen, Eugene, OR, USA) with 40,6-diamidino-2-phenylindole (2 lg/ml; Vector Laboratories, Burlingame, CA, USA). Images were captured using an LSM800 confocal microscope (Zeiss Laboratories, Thornwood, NY, USA). Z-stack images were processed with Volocity software for 3D reconstruction.
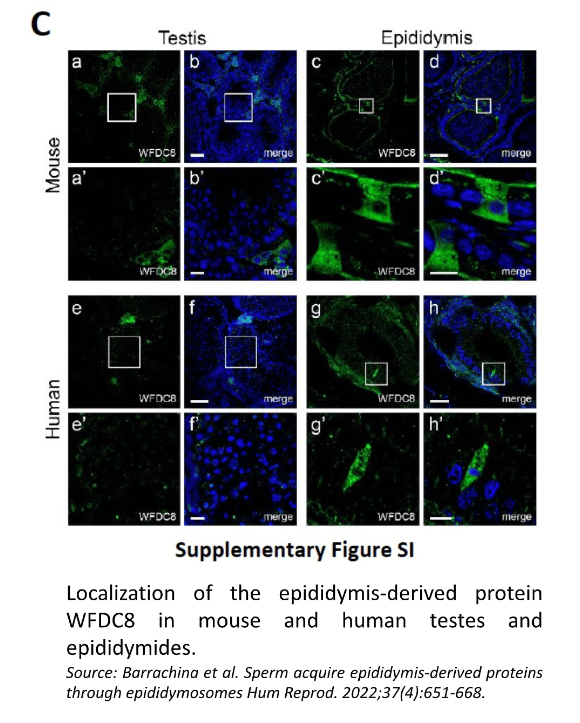
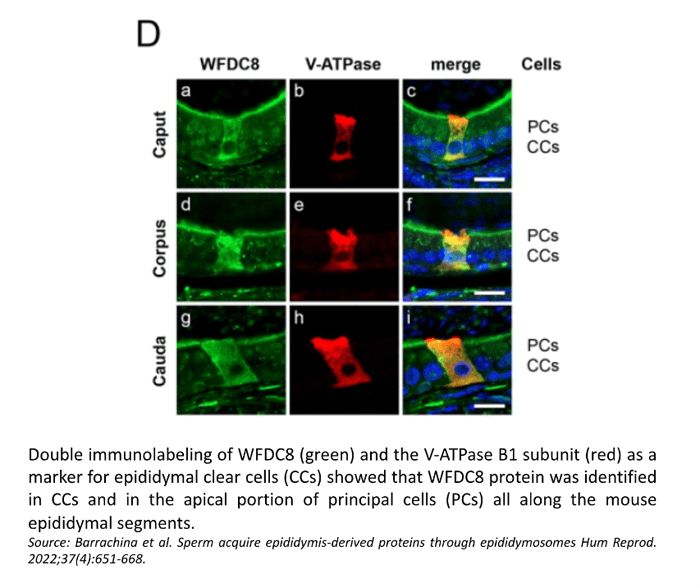
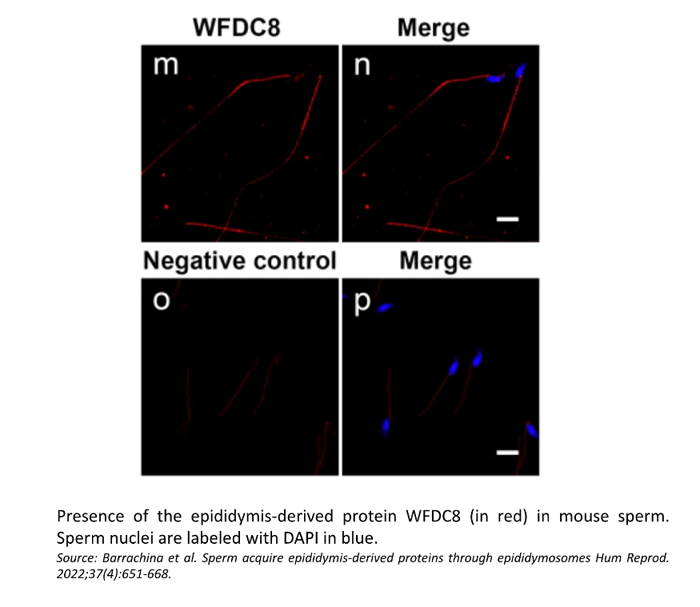
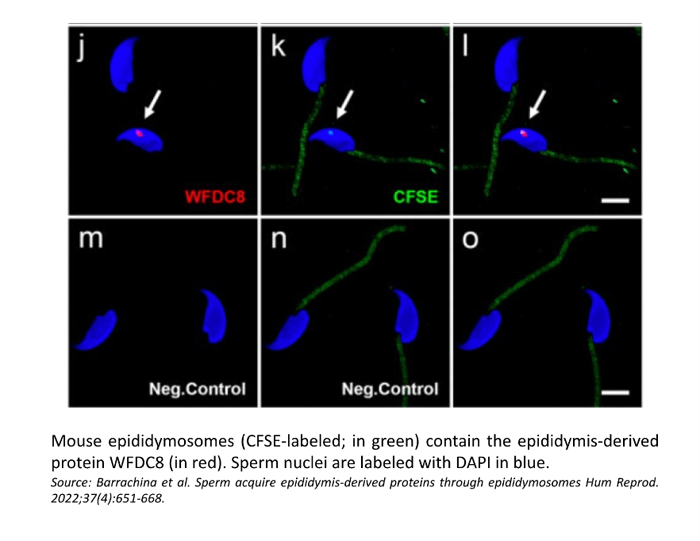
Confocal microscopy showed the presence of the WFDC8 protein in mouse and human epididymal epithelial cells, and the absence of WFDC8 in germ cells from seminiferous tubules of the testis (Figure 1). Double immunolabeling of WFDC8 and the V-ATPase B1 subunit as a marker for epididymal clear cells (CCs) showed that WFDC8 protein was identified in CCs and in the apical portion of principal cells all along the mouse epididymal segments (Figure 2). WFDC8 immunolabeling of mouse sperm showed that WFDC8 is present in the posterior part of the mouse sperm tail (Figure 3). We also demonstrated that mouse epididymosomes (extracellular vesicles from the epididymis) also contain the protein WFDC8 (Figure 4).
CUSABIO team. A Review of WFDC8 Antibody. https://www.cusabio.com/c-21085.html
Comments
Leave a Comment