Proteins are essential biomolecules involved in various biological processes, making their isolation and purification crucial for numerous applications in research, biotechnology, and medicine.
This article provides an in-depth exploration of protein purification, covering the definition and significance of protein purification, protein purification protocol, and methods for protein purification.
Table of Contents
Protein purification is a multi-step process aimed at obtaining a pure protein sample by isolating a specific protein of interest from a complex mixture of biomolecules, such as cellular lysates or culture supernatants.
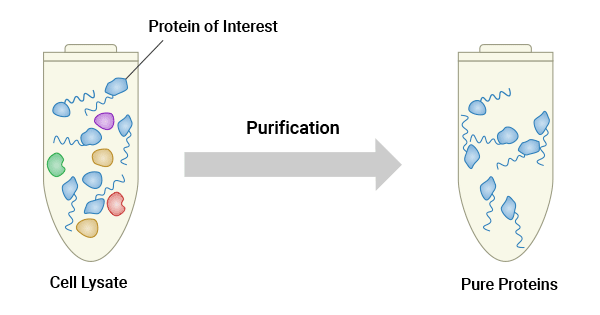
Figure 1. Protein purification
Protein purification is a fundamental step in biochemical and biomedical research, enabling the isolation, characterization, and utilization of proteins for diverse scientific and practical purposes.
Obtaining Pure Protein Samples: Pure proteins are essential for studying their structure and function, as well as protein-protein interactions accurately, minimizing the interference of contaminants to the experimental results.
Determination of Protein Structure: Purified proteins are essential for structural biology studies, including X-ray crystallography, nuclear magnetic resonance (NMR) spectroscopy, and cryo-electron microscopy (cryo-EM), providing high-resolution insights into protein structures and interactions.
Functional Studies: Purified proteins are necessary for elucidating their biochemical properties, such as enzymatic activity, binding affinities, and regulatory mechanisms, which are fundamental to unraveling biological processes.
Quality Control: Protein purification ensures the removal of contaminants and impurities, guaranteeing the integrity and purity of wanted proteins, which is necessary for the suitability of downstream applications and the reproducibility of experimental results.
Biotechnological Applications: Purified proteins are used in various biotechnological applications, including drug discovery, diagnostic assays, and biopharmaceutical production. Highly pure proteins are required for developing therapeutic agents and vaccines.
Disease Research: Purified proteins facilitate investigations into the pathogenesis of various diseases, biomarker discovery, and drug target identification, which is useful for developing therapeutic options for various diseases, including cancer, neurodegenerative disorders, and infectious diseases.
Generation of Antibodies: Pure proteins are used as immunogens to produce antibodies.
Protein purification typically involves several key steps, each aimed at separating the target protein from contaminants based on different physical and chemical properties.
Here is a general outline of the routine process of protein purification:
Cell Lysis
The process begins by breaking open cells to release the cellular contents, including the protein of interest. This can be achieved through mechanical disruption (e.g., sonication, homogenization), chemical methods (e.g., detergents, osmotic shock), or enzymatic digestion, depending on the type of cells and the desired protein.
Clarification
After cell lysis, the crude lysate is centrifuged or filtrated to separate insoluble cellular debris, such as cell membranes, organelles, and cell wall fragments, from the soluble protein fraction. The clarified supernatant contains the protein of interest along with various contaminants.
Capture
The clarified lysate is then subjected to a capture step, which aims to isolate, concentrate, and stabilize the target protein. It is a simple and rapid initial purification. This step usually uses affinity chromatography, where the resin is functionalized with ligands that interact with the target protein, such as affinity tags (e.g., His-tag, GST-tag) or antibodies.
Other protein purification assays are described in the following section.
Washing: Following capture, non-specifically bound proteins and contaminants are removed through a series of wash steps. These washes help to eliminate impurities while retaining the target protein bound to the resin.
Elution: The bound target protein is then eluted from the resin using elution buffers that disrupt the interactions between the protein and the ligand. Elution can be achieved through changes in pH, ionic strength, or
competitive displacement with excess ligands. The eluted protein is collected in fractions for further analysis and purification.
Polishing
Depending on the purity and activity requirements, additional purification steps may be necessary. These steps can include size exclusion chromatography to remove aggregates and contaminants based on size, ion exchange chromatography to further separate proteins based on charge, or other specialized techniques.
Quality Control
Throughout the purification process, quality control measures are essential to assess the purity, yield, and activity of the purified protein. Analytical techniques such as SDS-PAGE, Western blotting, mass spectrometry, and enzymatic assays are commonly used to evaluate the success of the purification and identify any remaining contaminants.
Concentration and Storage
Finally, the purified protein is concentrated to a desired concentration and stored under appropriate conditions to maintain its stability and activity.
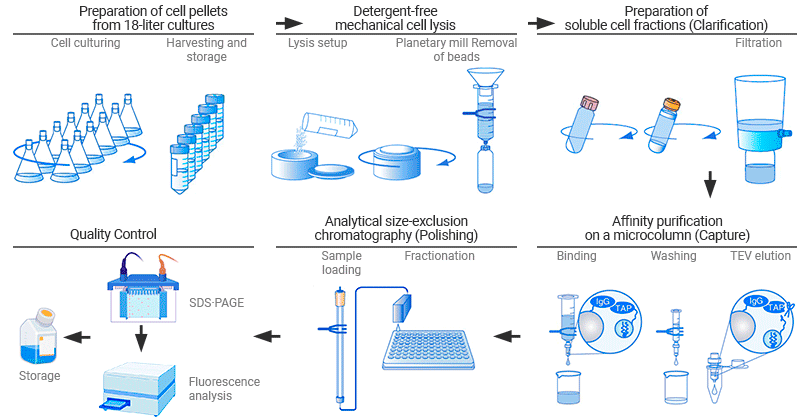
Figure 2. Protein purification procedures [7]
Protein purification is a complex process influenced by various factors that can impact the yield, purity, and activity of the final protein product. Optimizing protein purification requires careful consideration of numerous factors, including protein characteristics, sample complexity, purification methodology, experimental conditions, expression strategies, and quality control measures.
Expression Optimization: Appropriate expression host and expression conditions can affect protein yield, solubility, and post-modifications.
Method Selection: Choose appropriate purification methods based on protein characteristics (e.g. size, molecular weight, charge, solubility, and stability), sample complexity (e.g. cellular source and co-purifying substances such as nucleic acids, lipids, and other unwanted proteins), and downstream applications.
Buffer Condition Optimization: Buffer conditions such as pH, ionic strength, and additives, can affect protein stability, solubility, binding affinity, and elution efficiency. Optimizing these buffer conditions can enhance protein yield and purity.
Use of Appropriate Tags: Commonly used protein purification tags such as His-tags, GST-tags, or FLAG-tags can facilitate purification but may also affect protein structure and function. These tags are often co-expressed with recombinant proteins, facilitating the recombinant protein expression and purification.
Quality Control: Using analytical techniques such as SDS-PAGE, Western blotting, and mass spectrometry to assess protein purity and identify contaminants.
Protein purification employs a variety of techniques and methods, each designed to exploit different physical and chemical properties of proteins.
Table 1. Common protein purification techniques [1]
| Separation Mode | Separation basis | Mechanism | Resolving power | Advantages | Disadvantages | |
|---|---|---|---|---|---|---|
| Precipitation methods | Salting out | Solubility | Precipitation of proteins by reducing solubility with high salt concentrations, altering the hydration shell around proteins; often used as an initial step in protein purification to concentrate and partially purify proteins before further purification steps [2] | Low | Simple, rapid, cost-effective, scalability, | Low purity, loss of activity, limited applicability, time-consuming, may lead to protein denaturation |
| Organic solvent precipitation | Solubility | Addition of multifold organic solvent reduces the dielectric constant of the solution and increases the interaction between protein molecules, causing the significant reduction of protein solubility that leads to protein aggregation and precipitation | Low | High yields, can enrich low molecular weight proteins in soluble components, increase the detection coverage of small molecular weight proteins [3] | Complex, high cost, Organic solvents are toxic and flammable; | |
| Isoelectric point precipitation | Solubility, Charge properties | Precipitation of proteins at their isoelectric point, which has the lowest solubility | Low | Simple and rapid | Limited to proteins with distinct isoelectric points | |
| Nonionic polymer precipitation | Solubility | Precipitation of proteins with nonionic polymers such as dextran, NPEP, and polyethylene glycol (PEG) | Low | High yields, gentle purification | Requires careful optimization, may affect protein activity | |
| Centrifugation | Differential centrifugation | Shape, Size | Separation based on size and density differences, with centrifugation to separate components | Low, Medium | Relatively convenient to separate multiple samples simultaneously, effective for crude sample purification; not affect the cell viability and the distribution of hematopoietic cell populations [4] | Labor-intensive, limited resolution |
| Density gradient centrifugation | Size | Separation based on density gradients created by layers of solutions with different densities | Low, Medium | Can separate multiple components in mixed samples with high resolution and low cost | Time-consuming, gradient preparation | |
| Membrane protein purification | Dialysis protein purification | Size | Diffusion of small molecules across a semipermeable membrane, allowing separation of proteins from small molecules | Low, Medium | Gentle, suitable for small-scale purification | Slow process, limited to small proteins |
| Ultrafiltration | Size/Shape | Separation based on size exclusion using porous membranes that allow passage of molecules below a certain size | High | Rapid, efficient for concentrating proteins | Limited to proteins above a certain size | |
| Chromatography |
Gel filtration chromatography | Size | Separation based on molecular size using porous beads packed in a column | Medium | Suitable for the separation of proteins with larger or smaller molecular weight | Limited capacity, may require multiple steps |
| Ion exchange chromatography protein purification | Charge | Separation based on electrostatic interactions between proteins and charged groups on a resin | High | Versatile, applicable to a wide range of proteins | pH-dependent, may require optimization | |
| Hydrophobic interaction chromatography (HIC) | Hydrophobicity | Separation based on hydrophobic interactions between proteins and a hydrophobic ligand; mainly used to purify proteins sensitive to denaturation or high salt concentrations | High | High selectivity, suitable for protein purification | Protein denaturation, may require optimization | |
| Affinity chromatography protein purification | Specific affinity | Separation based on specific interactions between proteins and immobilized ligands with affinity for the protein of interest [5] | High | Fast, simple, and highly efficient; oftern used to isolate proteins with specific tags [6] | Costly, ligand immobilization required | |
| High-Performance Liquid Chromatography (HPLC) protein purification | Polarity, size, or charge | Separation based on properties like polarity, size, or charge using liquid chromatography columns | High | High resolution, automation, versatile | Expensive equipment, skilled operation required | |
Protein purification is a critical process that enables the isolation of pure and functional proteins for various applications in research and biotechnology. All protein purification methods have strengths and weaknesses. No single purification method can produce a 100% pure protein, but the combination of two or more methods can greatly improve the purity of the protein.
By understanding the principles behind protein purification, selecting appropriate techniques, and optimizing experimental conditions, researchers can achieve high-purity protein samples essential for advancing scientific knowledge and developing innovative biotechnological solutions.
Additional Considerations:
1. How to calculate percent yield for protein purification
The percent yield in protein purification is a crucial parameter that indicates the efficiency of the purification process. It provides valuable information about the success of isolating the target protein from the starting material.
A high percent yield indicates that a large proportion of the target protein was recovered during purification, signifying an effective and efficient purification method. On the other hand, a low percent yield suggests losses during the purification process, which may result from inefficiencies or limitations in the purification method.
Percent yield in protein purification can be calculated using the following formula:
Percent Yield = (Amount of purified protein obtained / Amount of protein in the starting material) × 100
2. how to calculate specific activity protein purification
The specific activity in protein purification is a key parameter that indicates the purity and functionality of the isolated protein. It represents the activity of the target protein per unit of total protein or per unit of sample volume.
A high specific activity indicates that a large proportion of the protein present in the sample is the desired protein with the intended biological activity, while a low specific activity suggests contamination or loss of activity due to incomplete purification or denaturation.
Specific activity in protein purification is calculated by dividing the activity of the purified protein (in units) by the total protein concentration (in mg).
Specific activity = Activity of purified protein / Total protein concentration
Further Reading:
Surface Markers of Mesenchymal Stem Cells
Stem Cell-What a Miraculous Resource in Human Body
Approaches to Determinate Protein Concentration
References
[1] Du, M., Hou, Z., et al. (2022). 1Progress, applications, challenges and prospects of protein purification technology [J]. Frontiers in Bioengineering and Biotechnology, 10.
[2] Duong-Ly K. C., Gabelli S. B. (2014). Salting out of proteins using ammonium sulfate precipitation [J]. Methods Enzymol. 541, 85–94.
[3] Periasamy P., Rajandran S., et al. (2021). A simple organic solvent precipitation method to improve detection of low molecular weight proteins [J]. Proteomics 21 (19), e2100152.
[4] Heib T., Gross C., Muller M. L., Stegner D., Pleines I. (2021). Isolation of murine bone marrow by centrifugation or flushing for the analysis of hematopoietic cells - a comparative study [J]. Platelets 32 (5), 601–607.
[5] Block H., Maertens B., et al. (2009). Immobilized-metal affinity chromatography (IMAC): A review [J]. Methods Enzymol. 463, 439–473.
[6] Lacki K. M., Riske F. J. (2020). Affinity chromatography: An enabling technology for large-scale bioprocessing [J]. Biotechnol. J. 15 (1), e1800397.
[7] Maeda, K., Poletto, M., Chiapparino, A., & Gavin, A. (2014). A generic protocol for the purification and characterization of water-soluble complexes of affinity-tagged proteins and lipids [J]. Nature Protocols, 9(9), 2256-2266.
Comments
Leave a Comment