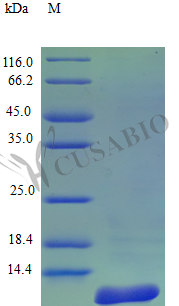
CCL8 (MCP-2) is a potent CC chemokine that recruits monocytes, T lymphocytes, and other immune cells through CCR1, CCR2B, and CCR5 signaling, making it a critical tool for dissecting leukocyte trafficking mechanisms. This tag-free recombinant protein encompasses the full-length mature sequence (24–99 aa) and demonstrates confirmed biological activity in chemotaxis bioassays using human peripheral blood monocytes at 10–100 ng/ml, providing a suitable basis for Transwell migration assays, monocyte activation studies, and downstream investigation of MAPK and NF-κB signaling pathways. Endotoxin levels below 1.0 EU/μg minimize LPS-driven artifacts that can confound cytokine-dependent readouts in primary immune cell experiments, while purity exceeding 96% by SDS-PAGE supports use as a reference standard in chemokine detection assays, a coating antigen for antibody validation by ELISA, or a defined stimulus in in vivo inflammation models.