Ovarian cancer, as a gynecological malignancy, exerts a significant impact on women's health. Its highly invasive nature and the challenge of early detection pose a threat to the quality and longevity of women's lives.
This article comprehensively explores the biological foundation, associated signaling pathways, clinical characteristics, treatment research, and the latest advancements in ovarian cancer. The aim is to foster a deeper understanding of ovarian cancer and its multifaceted aspects.
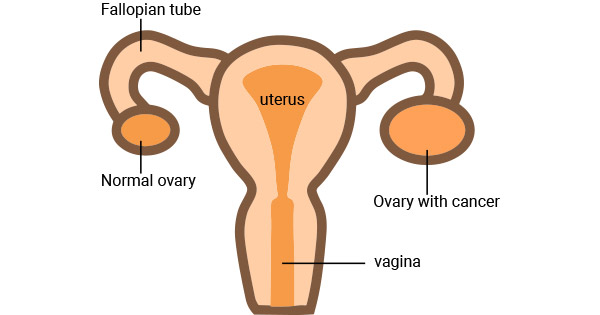
Figure 1. Ovarian cancer
1. Biological Basis of Ovarian Cancer
Ovarian cancer, as a complex and diverse malignancy, encompasses various aspects of biology, including classification, pathogenesis, pathophysiology, as well as molecular biology and genetics.
1.1 Classification
Ovarian cancer can be classified based on tissue type and cell origin. The main classifications include epithelial ovarian cancer, sex cord-stromal tumors, and germ cell tumors. Each type exhibits unique biological behaviors and clinical presentations.
Epithelial Ovarian Cancer: This is the most common type, including serous carcinoma, mucinous carcinoma, and endometrioid carcinoma, among others. Each subtype possesses distinct molecular characteristics and pathogenic mechanisms.
Sex Cord-Stromal Tumors: Originating from the connective tissue of the ovaries, tumors in this category exhibit different biological features and treatment approaches.
Germ Cell Tumors: Encompassing teratomas, embryonal carcinomas, etc., these tumors typically occur in young women, with molecular mechanisms and pathophysiology distinct from epithelial ovarian cancer.
1.2 Pathogenic Mechanisms
The pathogenic mechanisms of ovarian cancer involve complex interactions of various factors, including genetics, environment, hormones, and lifestyle. Familial genetic mutations, such as mutations in BRCA1 and BRCA2 genes, are closely associated with ovarian cancer. Additionally, chronic inflammation, infertility treatments, genetic susceptibility, among other factors, are considered significant contributors to ovarian cancer pathogenesis.
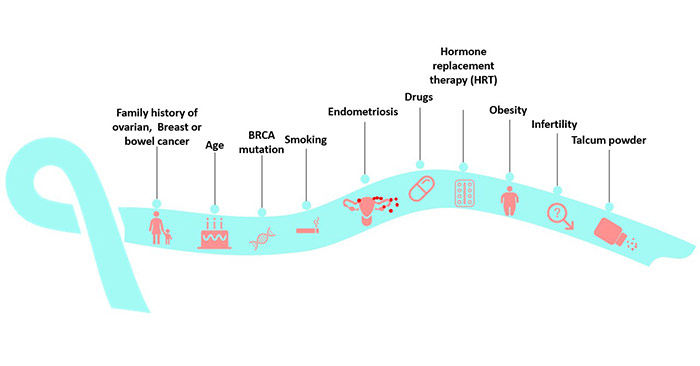
Figure 2. Risk factors for ovarian
1.3 Pathophysiology
Pathophysiological research focuses on aspects such as the growth, differentiation, angiogenesis, and invasion of ovarian cancer cells. Malignant growth of cancer cells leads to tumor formation, and the infiltration of tumor cells is one of the reasons for the highly invasive nature of ovarian cancer. Understanding these pathophysiological characteristics is crucial for devising effective treatment strategies.
1.4 Molecular Biology and Genetics
Studies in molecular biology and genetics have unveiled the molecular mechanisms of ovarian cancer, laying the foundation for precision medicine. Different types of ovarian cancer may exhibit distinct genetic variations and expression profiles. For instance, mutations in genes like BRCA1 and BRCA2 are associated with an increased risk of developing epithelial ovarian cancer. In-depth research in molecular biology aids in discovering new therapeutic targets and individualized treatment strategies.
In summary, the biological basis of ovarian cancer constitutes a complex and multi-layered system. A thorough understanding of these aspects contributes to a better comprehension of the pathophysiology and molecular mechanisms of ovarian cancer, paving the way for more precise treatment approaches for patients.
2. Ovarian Cancer-Related Signaling Pathways
A comprehensive understanding of the signaling pathways associated with ovarian cancer is crucial for unraveling its mechanisms of initiation and progression. Various signaling pathways exhibit abnormal activation, closely linked to the occurrence, development, and metastasis of ovarian cancer. Here are some key signaling pathways in ovarian cancer research:
The PI3K/AKT signaling pathway plays a pivotal role in the onset and progression of ovarian cancer. Its aberrant activation is associated with cellular processes such as survival, proliferation, growth, and metabolism. Excessive activation of the PI3K/AKT pathway in ovarian cancer is linked to increased tumor resistance and invasiveness. Inhibiting this pathway is considered a potential strategy for ovarian cancer treatment.
The RAS/MAPK signaling pathway participates in biological processes of ovarian cancer cells, including proliferation, differentiation, and metastasis. Mutations or overexpression of RAS protein family members can trigger abnormal activation of the MAPK pathway, contributing to the onset of ovarian cancer. Activation of this pathway is closely associated with the invasiveness and malignancy of ovarian cancer.
The Wnt/β-catenin signaling pathway is involved in cellular processes such as proliferation, differentiation, and self-renewal of stem cells. Aberrant activation of this pathway in ovarian cancer is associated with increased cancer stem cells and the invasiveness of cancer cells. Regulating the Wnt/β-catenin signaling pathway is considered a potential target for treating ovarian cancer.
The NOTCH signaling pathway plays a crucial role in various cancers, including ovarian cancer, by participating in processes such as proliferation, differentiation, and apoptosis. Abnormal activation of the NOTCH pathway is closely related to the occurrence and progression of ovarian cancer, making it a potential target for novel treatments.
The Hedgehog signaling pathway plays a vital role in embryonic development and adult tissue repair. In ovarian cancer, abnormal activation of this pathway is associated with the presence of cancer stem cells and tumor development. Therefore, the Hedgehog signaling pathway might be a potential target for ovarian cancer treatment.
The abnormal activation and interplay of these signaling pathways collectively contribute to the onset and progression of ovarian cancer. In-depth understanding of the molecular mechanisms of these pathways facilitates the discovery of new therapeutic targets, providing more possibilities for precision medicine and personalized treatments.
3. Ovarian Cancer-Related Biomarkers
Early diagnosis of ovarian cancer has been a challenging task, as symptoms often manifest in the late stages of the disease. Therefore, identifying reliable ovarian cancer biomarkers is crucial to enhance the accuracy and timeliness of early diagnosis. Here are some commonly used biomarkers in ovarian cancer research:
| Biomarker |
Function |
Applications |
Limitations |
| CA-125 |
Glycoprotein present in ovarian cancer tissue. |
Screening, diagnosis, and treatment monitoring of ovarian cancer.
Combined with other biomarkers or imaging techniques to improve diagnostic accuracy. |
Relatively low specificity and sensitivity.
Elevated in other conditions. |
| HE4 |
Member of the epidermal growth factor family, highly expressed in ovarian cancer tissue. |
Used in conjunction with CA-125 to enhance the diagnostic accuracy of ovarian cancer.
More sensitive in the early stages of ovarian cancer. |
Improved accuracy when used with CA-125 but not entirely specific.
Elevated in non-ovarian cancers. |
| AFP |
Fetal alpha-fetoprotein expressed during fetal development. |
Diagnosis of non-ovarian origin ovarian cancers, such as embryonal carcinoma.
Monitoring treatment effectiveness. |
Elevated in some non-hepatic conditions.
Limited specificity. |
| Inhibin |
Follicle-stimulating hormone inhibitor, may be elevated in ovarian cancer. |
Primarily used for the diagnosis of rare types of ovarian cancer, such as sex cord-stromal tumors.
Monitoring treatment effectiveness. |
Elevated in some non-ovarian tumors.
Limited specificity. |
The combination of these biomarkers and more advanced detection technologies holds the promise of improving early diagnosis levels of ovarian cancer in the future. However, further research and validation are still needed.
4. Latest Research Advances in Ovarian Cancer
Recent research highlights various aspects of ovarian cancer, including the relationship between gene mutations and tumor onset and drug resistance, prospective clinical models of epigenetic therapy, global epidemiology, and risk factors for ovarian cancer, the role of macrophages in the tumor microenvironment, the potential effects of natural compounds like quercetin in ovarian cancer treatment, the association between TAZ and ferroptosis, the regulatory role of circular RNA in ovarian cancer drug resistance, and more.
Gene Mutations and Drug Resistance: Mutations in epigenetic regulatory genes are closely associated with the occurrence and drug resistance of ovarian cancer [1]. Mutations leading to drug resistance pose a challenge in ovarian cancer treatment.
Prospective Epigenetic Therapy: Significant progress has been made in epigenetic therapy targeting ovarian cancer, providing new directions for future treatments [1].
Global Epidemiology of Ovarian Cancer: Global studies on the epidemiology and risk factors of ovarian cancer indicate an increasing incidence related to factors that elevate cancer risk
[2].
Macrophages in the Tumor Microenvironment: Polarization of macrophages in the tumor microenvironment plays a crucial role in ovarian cancer initiation, progression, and metastasis, offering new directions for macrophage-centered treatments [3].
Potential Role of Quercetin in Treatment: Quercetin, a natural compound, exhibits cytotoxic effects on ovarian cancer cells through various mechanisms, suggesting its potential as an ideal or adjunct agent in ovarian cancer treatment [4].
TAZ and Ferroptosis Relationship: The level of TAZ in chemo-resistant recurrent ovarian cancer is linked to reduced susceptibility to ferroptosis. TAZ promotes ferroptosis in ovarian cancers by regulating ANGPTL4 and NOX, providing a novel therapeutic potential [5].
Circular RNA Regulation of Drug Resistance: Abnormal expression of specific circular RNA (circCELSR1) is significantly associated with ovarian cancer drug resistance, offering new insights into the mechanisms of drug resistance [6].
Impact of the Tumor Microenvironment: The tumor microenvironment significantly influences the development, progression, prognosis, and treatment efficacy of ovarian cancer by modulating cancer hallmarks, providing insights for treatment strategy formulation [7].
Association of RPN2 Gene with Ovarian Cancer: Gene expression profile analysis reveals abnormal expression of the RPN2 gene in ovarian cancer, suggesting its association with ovarian cancer development [8].
References
[1] Sara Moufarrij, Monica Dandapani, Elisa Arthofer, Stephanie Gomez, et al. "Epigenetic Therapy for Ovarian Cancer: Promise and Progress", CLINICAL EPIGENETICS, 2019.
[2] Zohre Momenimovahed, Azita Tiznobaik, Safoura Taheri, Hamid Salehiniya, "Ovarian Cancer In The World: Epidemiology And Risk Factors", INTERNATIONAL JOURNAL OF WOMEN'S HEALTH, 2019.
[3] Huiyan Cheng, Zhichao Wang, Li Fu, Tianmin Xu, "Macrophage Polarization In The Development And Progression Of Ovarian Cancers: An Overview", FRONTIERS IN ONCOLOGY, 2019.
[4] Rana Shafabakhsh, Zatollah Asemi, "Quercetin: A Natural Compound For Ovarian Cancer Treatment", JOURNAL OF OVARIAN RESEARCH, 2019.
[5] Wen-Hsuan Yang, Zhiqing Huang, Jianli Wu, et al. "A TAZ–ANGPTL4–NOX2 Axis Regulates Ferroptotic Cell Death and Chemoresistance in Epithelial Ovarian Cancer", MOLECULAR CANCER RESEARCH, 2019.
[6] Shu Zhang, Jie Cheng, Chenlian Quan, et al. "CircCELSR1 (hsa_circ_0063809) Contributes to Paclitaxel Resistance of Ovarian Cancer Cells By Regulating FOXR2 Expression Via MiR-1252", MOLECULAR THERAPY. NUCLEIC ACIDS, 2019.
[7] Yuting Jiang, Chengdi Wang, Shengtao Zhou, "Targeting Tumor Microenvironment In Ovarian Cancer: Premise And Promise", BIOCHIMICA ET BIOPHYSICA ACTA. REVIEWS ON CANCER, 2020.
[8] Shahan Mamoor, "Differential Expression of Ribophorin II in Human Epithelial Ovarian Cancer", 2021.
CUSABIO team. A Comprehensive Overview of Ovarian Cancer. https://www.cusabio.com/c-20958.html





Comments
Leave a Comment