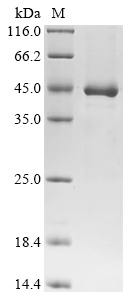
In order to produce the recombinant unknown prokaryotic organism phosphate-binding protein, the gene fragment encompassing the full-length protein (amino acids 1-376) and an N-terminal 10xHis-tag and C-terminal Myc-tag is cloned into a suitable expression vector. The cloning process involves inserting the gene fragment downstream of the appropriate promoter and regulatory elements. The resulting expression construct is then introduced into E. coli host cells through a transformation method. The transformed cells are selected to confirm the presence of the recombinant plasmid. Large-scale culture of the transformed E. coli cells is subsequently established in a growth medium optimized for protein expression. The presence of an N-terminal 10xHis-tag and C-terminal Myc-tag enables easy purification and detection of the recombinant phosphate-binding protein. Following cell lysis, the recombinant phosphate-binding protein is extracted and purified from the cell lysate. The purity of the recombinant phosphate-binding protein is evaluated through SDS-PAGE analysis, demonstrating a purity level of up to 90%. On the gel, the purified protein appears as a distinct band with an approximate molecular weight of 45 kDa.