Autosomal dominant polycystic kidney disease (ADPKD) is a slow, relentless disease: cysts expand, kidneys enlarge, and renal function declines over decades. Yet over the last few years, the therapeutic landscape has started to change, moving from pure supportive care to strategies that directly target cyst growth, metabolism, and even the underlying genetics. This article walks through those emerging approaches and how they might help us "rewrite" the natural history of ADPKD.
Table of Contents
1. Why Are New Treatments for ADPKD Needed?
ADPKD is the most common inherited kidney disease and a major cause of end‑stage kidney disease worldwide. Patients develop numerous fluid‑filled cysts in both kidneys, progressive kidney enlargement, hypertension, pain, and a range of extra‑renal problems such as liver cysts and intracranial aneurysms.
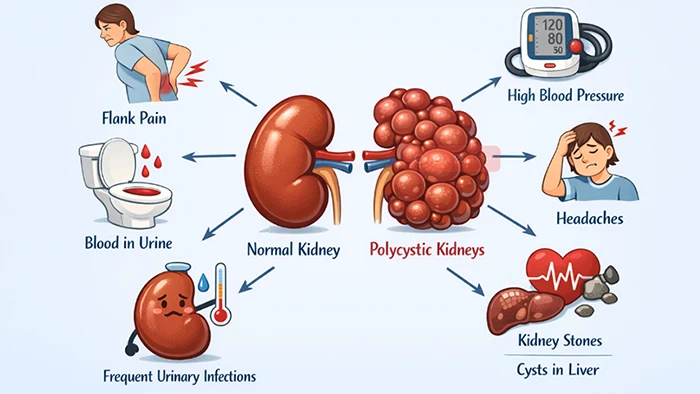
Figure. The diagram illustrat the symptoms of ADPKD
Standard care for ADPKD includes tight blood pressure control, often with renin–angiotensin–aldosterone system blockade, sodium restriction, pain and infection management, and cardiovascular risk reduction. These measures improve outcomes but do not directly slow cyst growth [1,2].
After decades of relying only on supportive measures, tolvaptan emerged as the first approved disease‑modifying therapy in ADPKD, but its benefit is modest and comes with significant trade‑offs.
Tolvaptan blocks the vasopressin V2 receptor (AVPR2) in the collecting duct, lowering cAMP levels and thereby reducing cyst epithelial proliferation and fluid secretion, and slowing the decline in estimated glomerular filtration rate (GFR). However, Tolvaptan therapy is limited by two major issues: V2 receptor blockade causes side effects like polyuria, thirst, and nocturia, compromising adherence and quality of life; and potential hepatotoxicity necessitates regular liver monitoring and can lead to treatment discontinuation [2,5].
Access and cost also limit implementation in many settings, and current indications focus on patients at higher risk of rapid progression, based on imaging, genotype, or clinical scores such as PROPKD. Together, these issues underline the need for additional, better‑tolerated, and more flexible disease‑modifying treatments that act on metabolism, fibrosis, inflammation, and gene‑level mechanisms [1,3,4,5].
2. Pathophysiological Basis for ADPKD's Targeted Therapy
To understand why these new therapies look promising, it helps to briefly review how gene defects trigger cyst growth. The key point is that polycystin dysfunction alters calcium and cAMP signaling, cell proliferation, and fluid secretion, creating multiple druggable pathways.
2.1 PKD genes and cystogenic signaling
Most cases of ADPKD are caused by mutations in PKD1 or PKD2, which encode polycystin‑1 (PC1) and polycystin‑2 (PC2). These proteins localize to primary cilia and other compartments in renal tubular epithelial cells and act as part of a mechanosensory and signaling complex. When PC1 or PC2 function is reduced, intracellular calcium signaling is altered, cAMP levels rise, and downstream pathways that drive proliferation and chloride‑dependent fluid secretion are activated [2,6,7].
The result is a transition of tubular epithelial cells into a cyst‑forming phenotype that both overproliferates and secretes fluid into the lumen. Over time, repeated cycles of cyst initiation and expansion, coupled with aberrant signaling, lead to kidney enlargement and structural distortion [2].
2.2 Key dysregulated pathways in ADPKD
Multiple signaling networks are dysregulated in ADPKD, and many are now being explored as therapeutic targets. These include:
- cAMP–PKA signaling, which promotes chloride secretion and proliferation in cystic epithelia [2].
- Growth and survival pathways such as the mTOR pathway, PI3K/AKT pathway, and MAPK/ERK pathway [2,3].
- Metabolic reprogramming, with shifts toward glycolysis and altered mitochondrial function, is reminiscent of a "Warburg‑like" phenotype [7,8].
- Pathways driving renal inflammation and interstitial fibrosis [2,7].
Because these pathways intersect, it is unlikely that a single target will be sufficient for most patients, which is why combination and staged approaches are increasingly discussed.
3. New Therapeutic Strategies for ADPKD
Beyond standard vasopressin inhibition, emerging strategies for ADPKD are expanding to target interconnected drivers of disease progression. This evolving therapeutic landscape includes repurposed small-molecule inhibitors, anti-fibrotic and anti-inflammatory agents, novel approaches targeting ciliary signaling defects, and even gene-based therapies. Rational combination regimens and precision medicine frameworks are now central to advancing a more multidimensional and potentially curative treatment paradigm.
3.1 Repurposed and small‑molecule pathway inhibitors
Many emerging strategies involve small molecules that target intracellular signaling or metabolism, often repurposed from other indications. These agents are attractive because their safety profiles may already be partly understood, and they can be layered into existing care.
Here, we focus on mTOR‑directed approaches, cAMP pathway modulation beyond vasopressin antagonism, and metabolic reprogramming.
3.1.1 mTOR and proliferation‑focused approaches
mTOR is a central regulator of cell growth and proliferation and is activated in cystic epithelia in ADPKD. Early preclinical work suggested that mTOR inhibitors such as sirolimus and everolimus could reduce cyst growth and protect kidney function in animal models [2,3].
However, clinical trials in humans showed mixed results. mTOR inhibitors reduced kidney volume in some studies but did not consistently slow GFR decline, and adverse effects, including mouth ulcers, dyslipidemia, and proteinuria. These experiences suggest that timing, dosing, and patient selection may need refinement, and that mTOR blockade alone is unlikely to be the full answer [2,3].
3.1.2 cAMP modulation beyond vasopressin antagonism
Because elevated cAMP is central to cyst growth, other ways to dampen this pathway have been explored. Somatostatin analogues can lower cAMP in cholangiocytes and renal epithelia and have shown some effect on liver cyst volume, but data on renal endpoints in ADPKD are less robust and somewhat variable [1,2].
Additional approaches, such as targeting phosphodiesterases that degrade cAMP, are being investigated in preclinical models. While these strategies are not as advanced as vasopressin antagonism, they may eventually serve as alternatives or adjuncts for patients who cannot tolerate tolvaptan [2,3].
3.1.3 Metabolic reprogramming and cell cycle targets
A growing body of work suggests that cystic epithelia undergo metabolic reprogramming, including increased glycolysis, altered oxidative phosphorylation, and disturbed mitochondrial function. This has led to interest in agents that modulate cellular metabolism [7,8].
Metformin, a widely used antidiabetic drug, activates AMP‑activated protein kinase (AMPK), which can inhibit mTOR, reduce proliferation, and improve mitochondrial health. In PKD rodent models, metformin slowed cyst expansion, reduced the kidney weight‑to‑body weight ratio, and improved kidney function and blood pressure. Early clinical trials such as TAME‑PKD have found metformin to be safe and well tolerated in non‑diabetic adults with ADPKD, with hints—but not yet definitive proof—of slower GFR decline [7-9].
Other metabolic strategies under investigation include ketogenic or caloric restriction‑like diets, SGLT2 (sodium‑glucose cotransporter 2) inhibitors, GLP‑1 receptor agonists, and lipid‑modifying agents like bempedoic acid. Many of these interventions are in phase 2 trials to exploit metabolic vulnerabilities in cystic cells while supporting overall kidney health [1,2].
3.2 Anti‑fibrotic, anti‑inflammatory, and microenvironment‑targeted therapies
Cyst growth does not happen in isolation. The surrounding kidney tissue becomes inflamed and fibrotic, and this microenvironment contributes to progression. Therapeutic strategies that address fibrosis and inflammation may therefore complement cyst‑directed approaches.
Emerging work in this area borrows heavily from broader chronic kidney disease (CKD) research and seeks to adapt it to the specific features of ADPKD.
3.2.1 Renal fibrosis and extracellular matrix remodeling
Interstitial fibrosis is a strong predictor of kidney function loss in ADPKD, as in other CKD forms. Fibroblasts, myofibroblasts, and altered extracellular matrix accumulate around expanding cysts, compressing remaining nephrons and blood vessels [2,3].
Anti‑fibrotic strategies under consideration include modulation of TGF‑β signaling, blockade of specific integrins, and agents that interfere with fibroblast activation and matrix deposition. While some of these therapies have been tested or approved in other fibrotic diseases, their application to ADPKD remains largely preclinical or early‑phase, and careful evaluation of risk–benefit balance will be essential [2,3].
3.2.2 Targeting inflammation and immune pathways
Cystic kidneys show infiltration by immune cells and increased levels of inflammatory cytokines and chemokines. These inflammatory signals may promote cyst growth and fibrosis, suggesting that targeted anti‑inflammatory therapies could be beneficial [2].
Candidate approaches include modulators of NF‑κB signaling, chemokine receptor antagonists, or more selective cytokine‑targeted drugs. However, long‑term systemic immunomodulation carries risks, so strategies that achieve local or pathway‑selective effects are of particular interest [2,3].
3.3 Cilia, mechanosensation, and novel mechanism‑based therapeutics
Because polycystins reside in primary cilia and other mechanosensory structures, ciliary biology has become a central focus in ADPKD research. Therapies that restore or compensate for defective ciliary signaling could tackle disease closer to its root.
These efforts span both pharmacologic approaches and gene‑ or RNA‑based strategies that directly address PKD gene function.
3.3.1 Restoring or compensating for ciliary signaling defects
Primary cilia integrate mechanical and chemical cues from tubular fluid flow and translate them into intracellular calcium and other signals. In ADPKD, defective polycystin complexes in cilia disrupt these signals and contribute to abnormal cAMP and growth pathways [2,6].
Potential therapeutic concepts include modulating calcium channels, downstream transcription factors, or other ciliary signaling components to normalize responses even in the context of polycystin deficiency. While still early, such "signal correction" strategies could complement more downstream pathway inhibitors [2].
3.3.2 Gene and RNA‑based approaches
Gene‑level therapies aim to correct or bypass the underlying PKD mutations. Recent advances in gene editing and RNA therapeutics have generated several lines of investigation:
- Antisense oligonucleotides targeting microRNAs such as miR‑17, which regulate networks involved in cyst growth [2,6].
- Viral or non‑viral delivery of Pkd1 or key functional fragments of PC1, such as its C‑terminal tail, which have shown cyst‑suppressing effects in mouse models [6].
- CRISPR–Cas9‑based approaches designed to repair or modulate PKD1 regulatory regions, although these are currently at a very early, preclinical stage [6].
These strategies have demonstrated promising effects in animal models but face substantial challenges, including safe delivery to the kidney, long‑term control of expression, off‑target effects, and cost. Nonetheless, they represent the first serious attempts to move from slowing the disease to potentially correcting its root molecular cause.
3.4 Combination and precision medicine strategies
Given the complex and interconnected pathways driving ADPKD, there is growing recognition that single‑agent therapy may not be enough for many patients. This has led to increased interest in rational combinations and precision medicine approaches.
The central idea is to match the right combination of therapies to the right patient at the right time, using robust biomarkers and risk‑stratification tools [2,3,10].
3.4.1 Rationale for combination therapy
Combining agents that act on different aspects of cyst biology—such as cAMP, metabolism, and fibrosis—could yield additive or synergistic effects on disease progression. For example, a vasopressin antagonist might be paired with a metabolic modulator like metformin, or an anti‑fibrotic agent, to simultaneously address cyst expansion, energy metabolism, and interstitial damage [2,3].
However, combination therapy also raises concerns about cumulative toxicity, adherence, and cost. This makes careful preclinical modeling, dose finding, and thoughtful trial design essential.
3.4.2 Biomarkers, risk stratification, and trial enrichment
Tools to predict progression and monitor treatment response are central to precision approaches. Height‑adjusted total kidney volume (htTKV), measured by MRI, is now widely used as a structural marker of disease burden and a surrogate for progression risk. Decline in estimated GFR remains a key functional endpoint [3,10].
Genotype, age at onset of hypertension or urologic events, and scoring systems such as PROPKD add further prognostic information. These tools enable risk stratification, support earlier intervention in high‑risk patients, and help enrich clinical trials for those most likely to show a treatment effect [3,10].
Going forward, molecular biomarkers derived from transcriptomics, proteomics, or metabolomics may further refine these classifications and guide therapy choices [2,3].
4. Translational and Clinical Trial Landscape of ADPKD
The ADPKD field is now in a phase where multiple new mechanisms are being translated from models into human studies. Understanding this landscape helps place individual therapies in context.
Overall, the trajectory is toward a more diverse toolbox of interventions that can be combined or sequenced according to disease stage and patient profile.
4.1 Key ongoing or recent clinical trials in ADPKD
Recent and ongoing trials in ADPKD include:
- Metformin trials (such as TAME‑PKD), evaluating safety, tolerability, and signals of efficacy in non‑diabetic adults with preserved GFR [7,8].
- Studies of SGLT2 inhibitors and GLP‑1 receptor agonists, leveraging their renal and metabolic benefits observed in diabetic and non‑diabetic CKD [1,2].
- Early‑phase evaluations of agents targeting inflammatory or fibrotic pathways, and of RNA‑based therapies such as miR‑17 antisense oligonucleotides [2,6].
Most of these trials use kidney volume, GFR slope, and safety as primary or key secondary outcomes, reflecting lessons learned from earlier tolvaptan studies.
4.2 Lessons from past failures and partial successes
The mixed clinical performance of mTOR inhibitors and the modest effect size of tolvaptan have taught the field several lessons. These include the need to:
- Intervene early enough in the disease course to influence long‑term trajectory.
- Select patients with a sufficiently high risk of progression to show a signal within realistic trial durations [10].
- Use doses and regimens that balance pathway inhibition with acceptable toxicity.
These experiences are now shaping the design of newer trials and encouraging more integrated, mechanism‑driven development strategies [3].
5. Future Directions and Research Opportunities of ADPKD
Looking ahead, ADPKD therapy is likely to become more personalized, more mechanism‑based, and more combinatorial. Several trends are converging to make this possible.
On the discovery side, multi‑omics and advanced models are deepening our understanding of disease heterogeneity. On the translational side, gene‑ and RNA‑based approaches are moving from concept to early clinical exploration [2,3,6].
5.1 Integrating multi‑omics and systems biology
Genomic, transcriptomic, proteomic, and metabolomic profiling of cystic kidneys and patient cohorts are revealing distinct molecular signatures that may define disease subtypes and new targets. Systems biology approaches that integrate these data with imaging and clinical phenotypes could help identify pathways most relevant in a given patient and predict response to specific interventions [2,3].
This type of integrative profiling may ultimately guide not only who to treat and when, but also which combination of therapies is most likely to work for a given molecular pattern [3].
5.2 Emerging technologies: organoids, microphysiological models, and imaging
Kidney organoids derived from pluripotent stem cells and microfluidic "kidney‑on‑a‑chip" systems that model tubular flow and ciliary signaling are increasingly used to study ADPKD. These models can recapitulate cyst formation in vitro and offer platforms for testing candidate therapies in a human genetic background [1,2].
Advanced imaging, including MRI‑based measures of kidney structure and novel functional imaging techniques, is also improving our ability to monitor disease and treatment response non‑invasively. Together, these tools may improve preclinical‑to‑clinical translation and reduce late‑stage failures [3,10].
5.3 From disease modification to disease interception
Most current trials target adults with established cyst burden and measurable decline in GFR or rapid kidney growth. In the future, there is interest in shifting to “disease interception”: intervening earlier in life, perhaps even before overt cysts or decline are evident, in genetically high‑risk individuals [3].
Such approaches raise important ethical, regulatory, and safety considerations, especially for gene‑based therapies or long‑term metabolic interventions. Still, if carefully designed, early interventions could offer the greatest chance to truly rewrite the course of ADPKD rather than merely slowing its progression [3,6].
Conclusion
ADPKD is driven by complex and interconnected pathways that regulate ciliary signaling, cAMP, metabolism, proliferation, inflammation, and fibrosis. For many years, clinicians could only manage complications and wait for the kidneys to fail. Today, tolvaptan provides a first step toward disease modification, and a diverse pipeline of new therapies from metabolic modulators to gene‑based approaches is emerging to complement and extend this effect.
The likely future of ADPKD care will be a combination of precision therapy guided by robust imaging and molecular biomarkers and supported by advanced models and trial designs. As basic scientists, clinicians, and industry partners continue to collaborate, the goal is not just to slow cyst growth but to truly "rewrite" the natural history of ADPKD for the next generation of patients.
References
[1] Zhou JX, Torres VE. Autosomal Dominant Polycystic Kidney Disease Therapies on the Horizon [J]. Adv Kidney Dis Health. 2023 May;30(3):245-260.
[2] Fang C, Norouzi S, Garimella PS. Therapies in autosomal dominant polycystic kidney disease: beyond tolvaptan [J]. Curr Opin Nephrol Hypertens. 2025 Sep 1;34(5):368-374.
[3] Chebib FT, Perrone RD. Drug Development in Autosomal Dominant Polycystic Kidney Disease: Opportunities and Challenges [J]. Adv Kidney Dis Health. 2023 May;30(3):261-284.
[4] Irazabal MV, Blais JD, et al. Prognostic Enrichment Design in Clinical Trials for Autosomal Dominant Polycystic Kidney Disease: The TEMPO 3:4 Clinical Trial [J]. Kidney Int Rep. 2016 Aug 6;1(4):213-220.
[5] Muto S, Okada T, et al. Long-term safety profile of tolvaptan in autosomal dominant polycystic kidney disease patients: TEMPO Extension Japan Trial [J]. Drug Healthc Patient Saf. 2017 Oct 25;9:93-104.
[6] Xue, C., Lv, J., et al. (2025). Gene therapy in polycystic kidney disease: A promising future [J]. Journal of Translational Internal Medicine, 12(6), 543.
[7] Nowak KL, Hopp K. Metabolic Reprogramming in Autosomal Dominant Polycystic Kidney Disease: Evidence and Therapeutic Potential [J]. Clin J Am Soc Nephrol. 2020 Apr 7;15(4):577-584.
[8] Gao, J., & Yu, X. (2025). Metabolic Reprogramming in Autosomal Dominant Polycystic Kidney Disease: Role in Cystogenesis and Novel Therapeutic Approaches [J]. Biomedicines, 13(7), 1596.
[9] Pastor-Soler NM, Li H, et al. Metformin improves relevant disease parameters in an autosomal dominant polycystic kidney disease mouse model [J]. Am J Physiol Renal Physiol. 2022 Jan 1;322(1):F27-F41.
[10] Cornec-Le Gall E, Blais JD, et al. Can we further enrich autosomal dominant polycystic kidney disease clinical trials for rapidly progressive patients? Application of the PROPKD score in the TEMPO trial [J]. Nephrol Dial Transplant. 2018 Apr 1;33(4):645-652.
CUSABIO team. Emerging Therapeutic Strategies for Autosomal Dominant Polycystic Kidney Disease (ADPKD). https://www.cusabio.com/c-20929.html




Comments
Leave a Comment