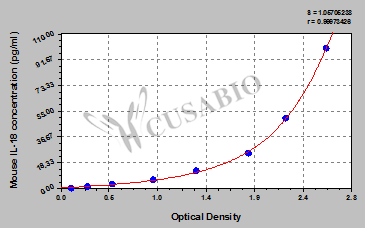
This mouse IL-18 ELISA kit uses the quantitative sandwich enzyme immunoassay technique to measure the levels of mouse IL-18 in the samples, including serum, plasma, cell culture supernates, and tissue homogenates. Antibody specific for IL-18 has been pre-coated onto the microplate. Standards and samples are pipetted into the wells and any IL-18 present is bound by the immobilized antibody. After removing any unbound substances, a biotin-conjugated IL-18 antibody is added to the wells. After washing, avidin conjugated Horseradish Peroxidase (HRP) is added to the wells, forming an antibody-antigen-enzyme-labeled antibody complex. Following a wash to remove any unbound HRP-avidin, the TMB substrate solution is added to the wells and the color develops into blue. The color changes from blue to yellow after adding the stop solution to the wells. The color intensity is proportional to the amount of IL-18 bound in the initial step.
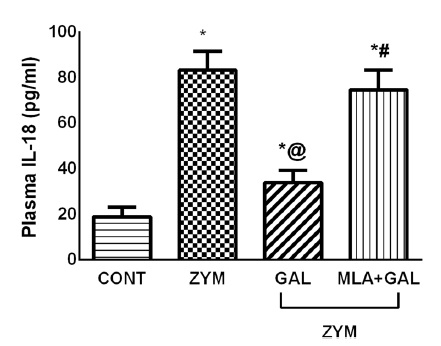
IL-18 is a powerful pro-inflammatory cytokine involved in the regulation of both innate and acquired immune responses. It plays a key role in autoimmune, inflammatory, and infectious diseases. IL‐18 binds to IL‐18Rα and IL‐18Rβ, activating NF‐κB and inducing inflammatory mediators such as adhesion molecules, chemokines, and cellular adhesion molecules. In certain cell types, IL18 also induces MAPK and PI3K/ATK signaling pathways resulting in the generation and release of pro-inflammatory cytokines. IL-18 is associated with obesity, insulin resistance, hypertension, and dyslipidemia. High expression of IL-18 has been found in many cancer types, including colorectal cancer, multiple myeloma, and pancreatic cancer, and has been associated with cell migration, invasion, proliferation, and metastasis of tumor cells.