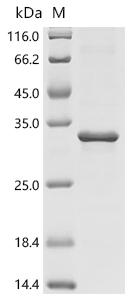
Recombinant Human Putative endogenous retrovirus group K member 11-1 Env polyprotein (ERVK11-1) gets expressed in E. coli and includes the complete sequence spanning amino acids 1 to 191. A C-terminal 6xHis-tag has been added to the protein, which makes purification and detection more straightforward. SDS-PAGE analysis shows purity levels above 85%, suggesting this preparation should work reliably for research purposes.
The ERVK11-1 Env polyprotein comes from an endogenous retrovirus group K that's already integrated into human DNA. Scientists find this protein intriguing because it may play a role in different cellular processes and carries evolutionary importance. Studying proteins like this one could help us understand how endogenous retroviral elements might influence human biology and what roles they potentially play in health and disease.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
The human ERVK11-1 is a viral envelope protein that typically requires complex folding, disulfide bond formation, and glycosylation for bioactivity (e.g., receptor binding or membrane fusion). E. coli, as a prokaryotic expression system, lacks eukaryotic chaperones and cannot perform glycosylation or proper disulfide bond formation for such proteins. The C-terminal His tag may not directly interfere with folding, but the absence of post-translational modifications and the prokaryotic environment make correct folding unlikely. Without experimental validation, the protein is probably misfolded and inactive. The high purity indicates low impurities, but does not guarantee native conformation.
1. Antibody Development and Immunological Studies
This recombinant ERVK11-1 protein can be used as an immunogen for generating antibodies targeting linear epitopes, as antibody production often tolerates some misfolding. The His tag facilitates purification and immobilization for ELISA-based screening. However, antibodies generated may not recognize native, correctly folded ERVK11-1 in physiological contexts (e.g., in human tissues) due to conformational differences. Validate antibody specificity against native ERVK11-1 or full-length proteins from eukaryotic expression systems.
2. Protein-Protein Interaction Studies
Use with caution. The His tag enables pull-down assays, but if the protein is misfolded, interactions may be non-physiological or tag-mediated. Any identified binding partners must be validated using native ERVK11-1 (e.g., from mammalian cells) to ensure relevance. Without a folding confirmation, results could be misleading.
3. Biochemical Characterization and Stability Studies
Suitable for basic biophysical analyses (e.g., circular dichroism for secondary structure, dynamic light scattering for aggregation state). However, data may not reflect the native protein's properties due to potential misfolding and lack of glycosylation. Avoid extrapolating to functional mechanisms without activity validation.
4. ELISA-Based Detection Assay Development
This protein can be used as an antigen in ELISA to detect ERVK11-1-specific antibodies, as linear epitopes may be accessible. However, if the protein is misfolded, it may not accurately mimic native ERVK11-1, leading to false negatives/positives in clinical samples. Validate assays with native proteins or patient sera for reliability.
Final Recommendation & Action Plan
Before using this recombinant ERVK11-1 for any application, prioritize experimental validation of its folding and potential bioactivity. Start with biophysical assays (e.g., circular dichroism to check secondary structure, size-exclusion chromatography to monitor oligomerization) to assess folding status. If possible, perform functional tests (e.g., receptor-binding assays) though bioactivity is unlikely in E. coli-expressed protein. For antibody development, proceed but validate antibodies against eukaryotic-expressed ERVK11-1. Avoid interaction studies without folding confirmation. For reliable results, consider expressing ERVK11-1 in a eukaryotic system (e.g., mammalian cells) to ensure proper folding and modifications. Always include appropriate controls to account for folding limitations.