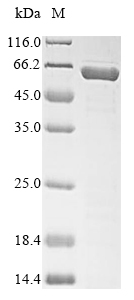
Recombinant Mouse Serine protease inhibitor A3N (Serpina3n) is expressed in E.coli and features a full-length mature protein spanning amino acids 21 to 418. It comes with an N-terminal 6xHis-SUMO tag, which helps with purification and detection. The product shows purity greater than 85% as determined by SDS-PAGE. This is strictly for research use only and is not suitable for therapeutic applications.
Serine protease inhibitor A3N, also known as Serpina3n, belongs to the serpin family. These proteins appear to play crucial roles in regulating protease activity. This particular protein acts as an inhibitor of serine proteases, which are involved in various biological processes such as inflammation and immune response. The way it modulates protease activity may make it a valuable tool for studying protease-related pathways and mechanisms in research settings.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
Recombinant Mouse Serine protease inhibitor A3N (Serpina3n) is expressed in E.coli and features a full-length mature protein spanning amino acids 21 to 418. It comes with an N-terminal 6xHis-SUMO tag, which helps with purification and detection. The product shows purity greater than 85% as determined by SDS-PAGE. This is strictly for research use only and is not suitable for therapeutic applications.
Serine protease inhibitor A3N, also known as Serpina3n, belongs to the serpin family. These proteins appear to play crucial roles in regulating protease activity. This particular protein acts as an inhibitor of serine proteases, which are involved in various biological processes such as inflammation and immune response. The way it modulates protease activity may make it a valuable tool for studying protease-related pathways and mechanisms in research settings.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
The mouse Serpina3n protein is expressed in E. coli, a prokaryotic system that often fails to properly fold eukaryotic proteins, especially those like Serpina3n that may require specific post-translational modifications, disulfide bond formation, or chaperone-assisted folding for native conformation. While the N-terminal 6xHis-SUMO tag can improve solubility and potentially aid folding, no functional assays (e.g., serine protease inhibition activity) or structural validation (e.g., circular dichroism) are mentioned. Thus, the protein's folding and activity status are uncertain without experimental verification.
1. Protein-Protein Interaction Studies Using Pull-Down Assays
The N-terminal 6xHis-SUMO tag facilitates purification and immobilization for pull-down experiments. However, if the protein is not correctly folded, it may not retain native binding properties, leading to false positives or negatives. Researchers should first validate folding (e.g., via activity assays) before use. The SUMO tag could enhance stability, but results should be interpreted cautiously.
2. Antibody Development and Validation
This recombinant Serpina3n can serve as an immunogen, but if misfolded, generated antibodies may not recognize the native protein in physiological contexts. The high purity (>85%) reduces contamination risks, but antibodies should be validated against correctly folded Serpina3n. Techniques like ELISA or Western blotting must include controls for conformational specificity.
3. Structural and Biophysical Characterization Studies
These applications require a correctly folded protein. If misfolded, structural data (e.g., from crystallography or NMR) would be invalid. Biophysical assays (e.g., circular dichroism) should first confirm folding. The E. coli expression system may produce aggregates or non-native conformations, limiting reliability without validation.
4. In Vitro Biochemical Assays and Enzyme Kinetics
This application depends on bioactivity. Without evidence of correct folding, inhibitory activity against serine proteases may be absent. Researchers should perform preliminary activity tests before kinetics studies. If inactive, the protein is unsuitable for such assays.
5. Cell-Based Functional Studies
Adding recombinant protein to cells assumes bioactivity and proper folding. If misfolded, cellular responses may be non-physiological. The His-SUMO tag aids tracking, but functionality must be verified (e.g., via protease inhibition in cells). Dose-response studies should include folding controls.
Final Recommendation & Action Plan
Given the uncertainty in protein folding and bioactivity, it is essential to prioritize validation before any application. Recommend performing functional assays (e.g., testing serine protease inhibition activity) and structural checks (e.g., circular dichroism or size-exclusion chromatography) to confirm native conformation. If validated, the protein can be used as described; otherwise, applications like enzymatic studies or cellular assays should be avoided or reinterpreted. Always include appropriate controls and consider using alternative expression systems (e.g., mammalian cells) if folding issues persist.