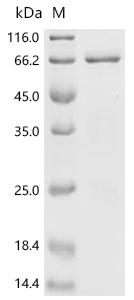
Recombinant Xenopus laevis Bic-C protein is produced using an E.coli expression system and includes an N-terminal 6xHis-SUMO tag. The protein covers amino acids 2 to 505, representing a partial sequence that appears useful for various research applications. Purity levels exceed 85% according to SDS-PAGE verification, which likely ensures reliable experimental results.
Bic-C protein in Xenopus laevis plays a recognized role in RNA binding and regulation. This function seems essential for understanding post-transcriptional gene expression mechanisms. The protein participates in pathways connected to mRNA localization and stability, positioning it as an important target for cellular and developmental biology studies. Researchers often turn to this protein when investigating RNA dynamics and interactions within these pathways.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
Based on the provided information, the recombinant Xenopus laevis Bic-C protein is expressed in E. coli, a prokaryotic system that may not support proper folding of eukaryotic proteins, especially those with complex structures or post-translational modifications. Bic-C is an RNA-binding protein involved in developmental processes, and its functionality likely depends on correct folding for interactions with RNA or other proteins. The protein is expressed as a partial fragment (2-505aa), which may lack full functional domains, and the N-terminal 6xHis-SUMO tag could potentially interfere with native folding despite improving solubility. Since activity is unverified, the protein cannot be assumed to be correctly folded or bioactive. While the SUMO tag may aid folding, experimental validation is essential to confirm conformation and function.
1. Protein-Protein Interaction Studies Using Pull-Down Assays
The N-terminal 6xHis-SUMO tag allows for efficient purification and immobilization, but if the Bic-C protein is misfolded, it may not interact authentically with binding partners from Xenopus laevis lysates, leading to non-physiological results. The partial length (2-505aa) limits interactions to domains within this region, but folding must be validated first. If correctly folded, this application is feasible; otherwise, data may be misleading.
2. Antibody Development and Validation
This application is generally suitable. The recombinant Bic-C protein can serve as an immunogen for antibody production, as antibodies often recognize linear epitopes less dependent on folding. The high purity (>85%) and tags facilitate purification and screening in ELISA or Western blot. However, if misfolded, antibodies may not recognize the native protein in physiological contexts, so validation against full-length, correctly folded Bic-C is recommended.
3. Biochemical Characterization and Stability Studies
This application is appropriate and should be prioritized to assess folding. Techniques like size exclusion chromatography and dynamic light scattering can directly evaluate oligomerization and aggregation, providing insights into protein stability. The SUMO tag may influence behavior, but these studies are valuable even if the protein is misfolded, as they characterize the recombinant product.
4. Tag-Based Purification Method Development
This application is low-risk and correct, as it relies on the tags rather than protein function. The dual-tag system makes the protein a useful model for optimizing nickel-affinity chromatography or SUMO protease cleavage protocols. Purity >85% supports method development, and folding status is largely irrelevant here.
Final Recommendation & Action Plan
Given the uncertainty in folding, it is crucial to first perform biochemical and biophysical characterization (e.g., circular dichroism for secondary structure, size-exclusion chromatography for oligomeric state) to assess the protein's conformation. If resources allow, functional assays (e.g., RNA-binding tests) should be conducted to confirm bioactivity. If the protein is correctly folded, it can be used for interaction studies and antibody validation with native controls; if misfolded, focus on applications like antibody development (for linear epitopes) or tag-based purification methods. Always include appropriate controls and consider alternative expression systems (e.g., insect or mammalian cells) if folding issues persist for functional studies.