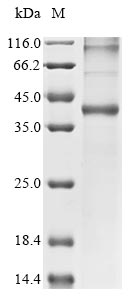
Recombinant Culex quinquefasciatus Odorant receptor (6031407) is produced through an in vitro E.coli expression system, covering the complete expression region of 1-393 amino acids. The protein carries an N-terminal 10xHis tag, which helps with purification and detection processes. Purity levels exceed 85% as determined by SDS-PAGE analysis. This product is designed strictly for research purposes and appears to deliver consistent performance in experimental settings.
The Odorant receptor from Culex quinquefasciatus likely plays a central role in how this mosquito species processes smell. It helps detect volatile compounds in the surrounding environment. This protein seems integral to signaling pathways that allow mosquitoes to respond to chemical cues, which makes it an interesting target for studying mosquito behavior and developing vector control strategies. Research on this receptor may contribute to our understanding of how smell works in insects and could lead to new pest management approaches.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
Odorant receptors are multi-transmembrane GPCR-like proteins that normally fold and function within the lipid bilayer of cell membranes. Expression in E. coli, especially in an in vitro cell-free system, almost always results in misfolding or aggregation of such hydrophobic membrane proteins unless they are refolded using detergents or reconstituted into liposomes or nanodiscs. Therefore, this recombinant odorant receptor is highly unlikely to fold correctly or possess native bioactivity when expressed in vitro E. coli without refolding steps. It can be used for immunogen preparation, antibody development, or linear epitope mapping, but not for functional or interaction assays involving ligand or co-receptor binding unless properly refolded into a membrane-mimetic system.
1. Antibody Development and Validation
The recombinant protein is well-suited as an immunogen for developing antibodies against Culex odorant receptors, since it represents the full-length sequence and maintains high purity (>85%). Even though it is not folded correctly, it will expose multiple linear epitopes, making it appropriate for generating antibodies useful in Western blotting, ELISA, or immunocytochemistry.
If properly refolded, the protein could additionally serve in conformational epitope validation for native-state receptor studies.
2. Biochemical Characterization and Structural Studies
The recombinant odorant receptor can be used for basic biochemical characterization, such as SDS-PAGE analysis, domain mapping, or limited proteolysis, but not for structural or biophysical studies unless refolded into a suitable membrane-mimetic environment. If detergent refolding is achieved, circular dichroism (CD) or fluorescence spectroscopy could provide insights into secondary structure content and folding efficiency. In its current form, studies should focus on analytical verification (mass spectrometry, immunoblotting) rather than native structural analysis.
3. Comparative Olfactory Receptor Research
The protein can still be used in comparative studies at the sequence or antibody recognition level. For example, antibodies raised against this recombinant receptor can help assess cross-reactivity across mosquito species. However, functional comparisons (e.g., odorant ligand binding) require receptors that are correctly folded in eukaryotic or membrane systems, such as insect cells or Xenopus oocytes.
4. In Vitro Assay Development
While the recombinant protein can act as a standardized reagent for assay development, it should not be used as a functional control for ligand binding or odorant detection assays.
Instead, it can serve as a non-functional calibration standard for optimizing purification, detection, or refolding protocols. If refolding is achieved, the protein can then be validated for functional screening purposes.
Final Recommendation & Action Plan
The vitro E. coli–expressed full-length Culex quinquefasciatus odorant receptor is structurally misfolded and biologically inactive under normal in vitro bacterial expression conditions, given its multi-transmembrane nature and dependence on lipid membranes for correct conformation. Therefore, this recombinant protein is best suited for antibody generation, epitope mapping, comparative immunological studies, and biochemical verification. It is not appropriate for functional assays, ligand binding, or receptor-cofactor interaction studies unless specifically refolded or reconstituted into lipid-based systems.