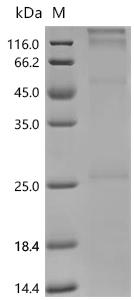
Recombinant Human Clarin-1 (CLRN1) is produced using an E.coli-based cell-free expression system and contains the full-length protein from amino acids 1 to 232. The protein has been engineered with an N-terminal 6xHis-tag to aid in purification, reaching a purity level greater than 85% as determined by SDS-PAGE. The protein features a four transmembrane domain structure and is presented using a detergent platform to maintain stability and functionality for research applications.
Clarin-1 appears to be a transmembrane protein that plays an important role in sensory perception, particularly within the auditory system. It seems to be involved in key cellular processes that may contribute to hearing function and maintenance. Understanding how Clarin-1 is structured and how it interacts with other components could be crucial for exploring its contributions to sensory pathways. This protein might also have potential implications in auditory research, as researchers study its impact on cellular signaling and sensory transduction.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
Based on the provided information, the recombinant Human CLRN1 is expressed using an in vitro E. coli expression system (cell-free system), which typically provides better folding conditions for membrane proteins by reducing cellular stress and aggregation. CLRN1 is a transmembrane protein with 4 transmembrane domains, making its proper folding challenging. The protein is full-length (1-232aa) with an N-terminal 6xHis tag, and purity is >85% by SDS-PAGE. However, since activity is unverified, the protein cannot be assumed to be correctly folded or bioactive. While the cell-free system improves folding probability, experimental validation is essential to confirm both proper folding (e.g., via circular dichroism or size exclusion chromatography) and bioactivity (e.g., through interaction assays with known binding partners).
1. Membrane Protein Biochemical Characterization Studies
This application is appropriate and should be prioritized. The recombinant CLRN1 can be used to study biochemical properties such as stability, detergent compatibility, and oligomerization state using techniques like size exclusion chromatography and dynamic light scattering. These studies can directly assess the protein's folding quality and are valuable even if the protein is not fully functional. The cell-free expression system provides a consistent protein preparation for reproducible characterization.
2. Antibody Development and Validation
This application is well-supported. The recombinant CLRN1 can serve as an effective immunogen for generating antibodies, as antibodies may recognize linear epitopes even if the protein is misfolded. The high purity (>85%) and His-tag facilitate purification and screening. However, if CLRN1 is misfolded, antibodies may not recognize conformational epitopes of the native protein. Validation against endogenous CLRN1 from mammalian cells is recommended to ensure physiological relevance.
3. Protein-Protein Interaction Studies
This application is feasible but requires caution. The His-tag enables pull-down assays to identify binding partners, but if CLRN1 is misfolded, interactions may be non-physiological. The 4-transmembrane domain structure is particularly sensitive to proper folding for native interactions. This application should only be pursued after confirming folding (e.g., via conformational antibodies or functional assays). Detergent solubilization may also alter some native interactions.
4. Membrane Protein Reconstitution Experiments
This application is high-risk without folding validation. While the detergent-solubilized CLRN1 can be reconstituted into lipid bilayers, if the protein is misfolded, its membrane integration behavior may not reflect native topology or function. Lipid composition studies may yield misleading data if the protein isn't properly structured. Folding should be confirmed before attempting reconstitution studies aimed at understanding biological mechanisms.
Final Recommendation & Action Plan
Given the uncertainty in folding and bioactivity, the recommended approach is to first perform comprehensive biochemical characterization (Application #1) to assess the protein's folding state, oligomerization, and stability. Techniques should include size exclusion chromatography (to check monodispersity), circular dichroism (for secondary structure), and thermal stability assays. If the protein shows proper folding, proceed with limited validation of protein-protein interactions using known binding partners. Antibody development (Application #2) can proceed concurrently, as it is less dependent on native conformation. Membrane reconstitution studies (Application #4) should be deferred until folding is validated. Always include appropriate controls, such as Western blotting with conformational antibodies when available, and compare results with literature on CLRN1 structure-function relationships where possible.