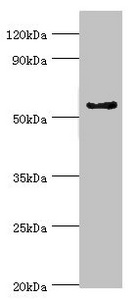
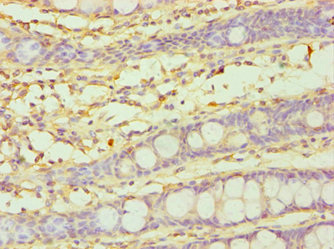
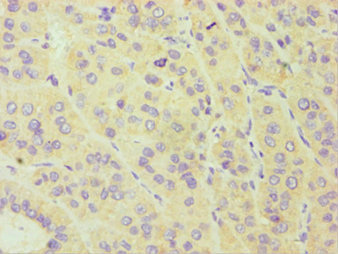
A cytochrome P450 monooxygenase involved in the metabolism of steroid hormones and vitamins. Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase). Catalyzes the hydroxylation of carbon-hydrogen bonds. Exhibits high catalytic activity for the formation of catechol estrogens from 17beta-estradiol (E2) and estrone (E1), namely 2-hydroxy E1 and E2. Catalyzes 6beta-hydroxylation of the steroid hormones testosterone, progesterone, and androstenedione. Catalyzes the oxidative conversion of all-trans-retinol to all-trans-retinal, a rate-limiting step for the biosynthesis of all-trans-retinoic acid (atRA). Further metabolizes all trans-retinoic acid (atRA) to 4-hydroxyretinoate and may play a role in hepatic atRA clearance. Also involved in the oxidative metabolism of xenobiotics, including calcium channel blocking drug nifedipine and immunosuppressive drug cyclosporine.