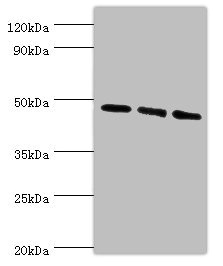
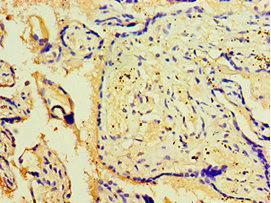
Component of the autophagy machinery that controls the major intracellular degradation process by which cytoplasmic materials are packaged into autophagosomes and delivered to lysosomes for degradation. Plays an important role in starvation- and calcium-mediated autophagy, as well as in mitophagy. Functions downstream of the ULK1 and PI3-kinases that produce phosphatidylinositol 3-phosphate (PtdIns3P) on membranes of the endoplasmic reticulum once activated. Binds phosphatidylinositol 3-phosphate (PtdIns3P), and maybe other phosphoinositides including PtdIns3,5P2 and PtdIns5P, and is recruited to phagophore assembly sites at the endoplasmic reticulum membranes. There, it assists WIPI2 in the recruitment of ATG12-ATG5-ATG16L1, a complex that directly controls the elongation of the nascent autophagosomal membrane. Together with WDR45/WIPI4, promotes ATG2 (ATG2A or ATG2B)-mediated lipid transfer by enhancing ATG2-association with phosphatidylinositol 3-monophosphate (PI3P)-containing membranes. Involved in xenophagy of Staphylococcus aureus. Invading S.aureus cells become entrapped in autophagosome-like WIPI1 positive vesicles targeted for lysosomal degradation. Plays also a distinct role in controlling the transcription of melanogenic enzymes and melanosome maturation, a process that is distinct from starvation-induced autophagy. May also regulate the trafficking of proteins involved in the mannose-6-phosphate receptor (MPR) recycling pathway.