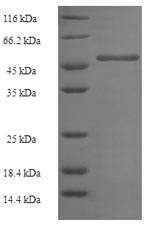
This Human GGPS1 recombinant protein was produced in E.coli, where the gene sequence encoding Human GGPS1 (1-300aa) was expressed with the N-terminal 6xHis-SUMO tag. The purity of this GGPS1 protein was greater than 90% by SDS-PAGE.
Geranylgeranyl pyrophosphate synthase (GGPS1) is a key enzyme in plant terpenoid biosynthesis. The enzyme catalyzes the synthesis of geranylgeranyl pyrophosphate (GGPP), which is a precursor to several holoterpenoids (carotenoids, kaurene, diterpenes) and meroterpenoids (side-chain of chlorophylls, tocopherols, plastoquinones, phylloquinones). The study identified a cDNA for the plastid-located GGPS1 from Capsicum annuum and showed a correlative increase in enzyme activity and transcript level during fruit ripening. The cloned cDNA codes for a high molecular weight precursor of 369 amino acids which contains a transit peptide of approximately 60 amino acids. In-situ immunolocalization experiments have demonstrated that geranylgeranyl pyrophosphate synthase is located exclusively in the plastids.
Reference:
Kuntz M, Römer S, Suire C, Hugueney P, Weil JH, Schantz R, Camara B. Identification of a cDNA for the plastid-located geranylgeranyl pyrophosphate synthase from Capsicum annuum: correlative increase in enzyme activity and transcript level during fruit ripening. Plant J. 1992 Jan;2(1):25-34.