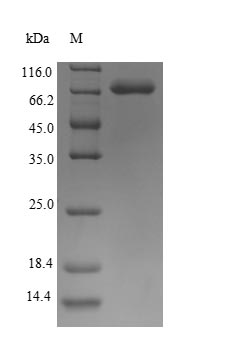
Recombinant Human Keratin, type I cytoskeletal 10 (KRT10) is produced in E. coli and contains the complete protein sequence spanning amino acids 1 to 584. The protein carries an N-terminal 6xHis tag, which makes purification and detection more straightforward. SDS-PAGE analysis shows the product achieves greater than 90% purity, suggesting it meets quality standards for research work.
Keratin, type I cytoskeletal 10, appears to be a structural protein with a key role in building and maintaining the cytoskeleton within epithelial cells. It mainly provides mechanical support and stability while helping to assemble intermediate filaments. Researchers studying skin biology and epithelial tissue often focus on this protein because its structural characteristics may offer important insights.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
KRT10 is a type I keratin that requires proper dimerization with type II keratins (e.g., KRT1) and specific post-translational modifications for its structural function in intermediate filament formation. The E. coli expression system lacks the eukaryotic machinery necessary for proper keratin folding, including molecular chaperones and modification systems. While the protein is full-length (1-584aa), the N-terminal 6xHis tag may interfere with the critical head domain structure essential for keratin-keratin interactions. Keratins require specific α-helical coiled-coil formation that is challenging to achieve in prokaryotic systems. The purity of >90% indicates minimal contaminants, but does not guarantee correct folding. Without validation (e.g., circular dichroism for α-helical content, co-sedimentation assays with type II keratins), the protein cannot be assumed to be properly folded or functional.
1. Protein-Protein Interaction Studies with Keratin Type II Partners
This recombinant KRT10 is unsuitable for interaction studies without folding validation. If misfolded, it will not form proper heterodimers with KRT1, leading to false negative results. The His-tag may sterically hinder the interaction interface in the head domain. Validate α-helical structure and dimerization capability before use, and consider tag removal for reliable interaction assays.
2. Antibody Development and Validation
This KRT10 protein can generate antibodies, but if misfolded, antibodies may recognize non-native epitopes rather than physiological KRT10 conformations. The His-tag may dominate the immune response. For specific antibodies, validate against native keratin filaments from human epithelial cells and use tag-free protein if possible. The high purity supports immunization, but doesn't ensure biological relevance.
3. Biochemical Characterization of Keratin Assembly Properties
The KRT10 protein cannot be used for filament assembly studies without demonstrating proper folding. Misfolded KRT10 will not polymerize correctly with type II keratins. The His-tag may disrupt assembly kinetics. First, validate secondary structure and dimer formation, then use eukaryotic-expressed keratins as positive controls for assembly experiments.
4. His-Tag Affinity-Based Binding Assays
While the His-tag enables technical applications like pull-down assays, identified interactions may be artifactual if the protein is misfolded. Use with caution and include rigorous controls (e.g., tag-only protein, properly folded keratin standards). For biological interactions, validate findings with native keratins.
Final Recommendation & Action Plan
This recombinant KRT10 requires extensive validation before use. Priority steps: 1) Verify secondary structure by circular dichroism (expecting high α-helical content); 2) Test dimerization capability with type II keratins using co-immunoprecipitation or size-exclusion chromatography; 3) Remove the His-tag if it interferes with functional domains; 4) Use mammalian-expressed KRT10 as a positive control for critical experiments. For antibody development, characterize sera against human skin sections. For interaction studies, confirm results with complementary methods using native keratins.