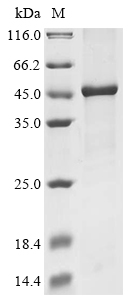
The recombinant Mouse Cyld was expressed with the amino acid range of 579-952. This Cyld protein is expected to have a theoretical molecular weight of 50.6 kDa. The Cyld protein was expressed in e.coli. The Cyld coding gene included the N-terminal 10xHis tag and C-terminal Myc tag, which simplifies the detection and purification processes of the recombinant Cyld protein in following stages of expression and purification.
The mouse ubiquitin carboxyl-terminal hydrolase CYLD is a deubiquitinating enzyme that can remove ubiquitin chains from target proteins, thereby controlling protein stability and cellular signaling pathways. One of its main functions is to negatively regulate NF-κB signaling by deubiquitinating key components in the pathway. CYLD also influences other cellular processes such as cell cycle progression, apoptosis, and immune response. Dysregulation of CYLD has been linked to various diseases, including cancers and inflammatory disorders. Understanding the molecular functions of CYLD provides insights into its potential therapeutic applications in these conditions.