HSP90 alpha and beta represent two of the most abundant and functionally critical molecular chaperones in eukaryotic cells, playing essential roles in protein folding, stability, and the maturation of key signaling molecules. These chaperones are indispensable for maintaining cellular proteostasis and have emerged as significant targets in cancer research, where they support the stability of numerous oncogenic client proteins. Understanding HSP90 biology provides crucial insights into stress response mechanisms, signal transduction pathways, and therapeutic vulnerabilities in disease states.
This recombinant monoclonal antibody, generated in rabbit and defined by clone 10D6, offers the consistency and reproducibility that demanding research applications require. Because recombinant antibodies are produced from a defined genetic sequence, researchers benefit from lot-to-lot uniformity that eliminates the variability often encountered with traditional hybridoma-derived reagents. This sequence-defined production ensures that experiments remain comparable across extended studies and between laboratories.
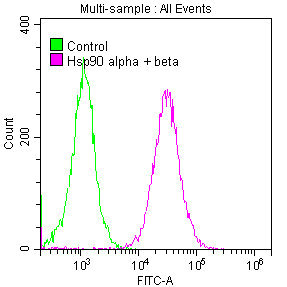
Validation data demonstrates robust performance across multiple experimental platforms. In western blotting, the antibody detects a clean 90 kDa band matching the predicted molecular weight across an extensive panel of human cell lines including HeLa, MCF-7, K562, HL-60, HepG2, A549, Jurkat, and PC-3, as well as rat brain tissue, confirming reliable cross-species reactivity. Immunoprecipitation studies in HeLa lysates show efficient target enrichment, while flow cytometry analysis in Jurkat cells demonstrates clear positive signal separation from control conditions.
With validated performance in ELISA, western blotting, flow cytometry, and immunoprecipitation, this antibody provides flexibility for researchers investigating signal transduction, stress response pathways, or chaperone-dependent mechanisms in both human and rat experimental systems.