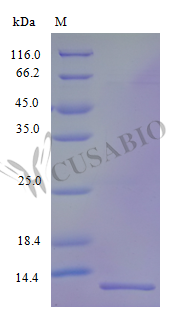
CXCL17, also known as VEGF coregulated chemokine 1, plays a distinctive role in mucosal immunity and angiogenesis-linked chemotaxis, making it a valuable tool for dissecting crosstalk between immune cell recruitment and vascular signaling. This tag-free recombinant human CXCL17 encompasses the full-length mature sequence (22–119 aa) expressed in *E. coli* and demonstrates an ED50 of less than 5.0 μg/ml for inducing VEGF expression in murine endothelial cells, corresponding to a specific activity exceeding 200 IU/mg — data that supports use in chemotaxis and migration assays, VEGF-dependent signaling pathway studies, and cell-based functional assays examining endothelial activation or macrophage recruitment. Endotoxin levels below 1.0 EU/μg minimize LPS-driven artifacts in sensitive immune cell experiments and in vivo inflammation models, while purity above 96% by SDS-PAGE provides a suitable basis for deployment as a reference standard in cytokine detection assays or as a reliable antigen for antibody development and ELISA validation.