[1] Lu, Chenglin, et al. "Research progress on the role of tumor‑associated macrophages in tumor development and their use as molecular targets." International Journal of Oncology 64.2 (2024): 1-12.
[2] Li, Xian, Jiaqi Peng, and Xiulan Su. "Expression of immune regulatory factors, chemokines and growth factors in differentiated gastric cancer cells treated with an anticancer bioactive peptide combined with oxaliplatin." Molecular and Clinical Oncology 20.1 (2024): 1-10.
[3] Chen, Xuyan, et al. "The role of CXCL chemokine family in the development and progression of gastric cancer." International journal of clinical and experimental pathology 13.3 (2020): 484.
[4] Mamazhakypov, Argen, et al. "The role of chemokines and chemokine receptors in pulmonary arterial hypertension." British journal of pharmacology 178.1 (2021): 72-89.
[5] Do, Ha Thi Thu, Chang Hoon Lee, and Jungsook Cho. "Chemokines and their receptors: multifaceted roles in cancer progression and potential value as cancer prognostic markers." Cancers 12.2 (2020): 287.
[6] Wasilko, David Jonathan, et al. "Structural basis for chemokine receptor CCR6 activation by the endogenous protein ligand CCL20." Nature communications 11.1 (2020): 3031.
[7] Martina, Maria Grazia, et al. "Discovery of small-molecules targeting the CCL20/CCR6 axis as first-in-class inhibitors for inflammatory bowel diseases." European Journal of Medicinal Chemistry 243 (2022): 114703.
[8] Isaikina, Polina, et al. "Structural basis of the activation of the CC chemokine receptor 5 by a chemokine agonist." Science Advances 7.25 (2021): eabg8685.
[9] Gómez-Melero, Sara, and Javier Caballero-Villarraso. "CCR6 as a Potential Target for Therapeutic Antibodies for the Treatment of Inflammatory Diseases." Antibodies 12.2 (2023): 30.
[10] Meitei, Heikrujam Thoihen, Nandadeep Jadhav, and Girdhari Lal. "CCR6-CCL20 axis as a therapeutic target for autoimmune diseases." Autoimmunity reviews 20.7 (2021): 102846.
[11] Reichard, Sherwood. The International Cytokine Conference (11th) Held in Dublin (Ireland) on September 20-24 2003 (European Cytokine Network, Volume 14, Number 3, September 2003). 2003.
[12] Lee, Adrian YS, et al. "The relationship between CCR6 and its binding partners: Does the CCR6–CCL20 axis have to be extended?." Cytokine 72.1 (2015): 97-101.
[13] Liu, Kaiwen, et al. "Structural insights into the activation of chemokine receptor CXCR2." The FEBS Journal 289.2 (2022): 386-393.
[14] Murugesan, Vidya, and Senthilkumar Rajagopal. "Modulatory effects of G protein–coupled receptor in hepatocellular carcinoma." Theranostics and Precision Medicine for the Management of Hepatocellular Carcinoma. Academic Press, 2022. 81-95.
[15] Liu, Jinlin, et al. "Tumor-associated macrophages recruit CCR6+ regulatory T cells and promote the development of colorectal cancer via enhancing CCL20 production in mice." PloS one 6.4 (2011): e19495.
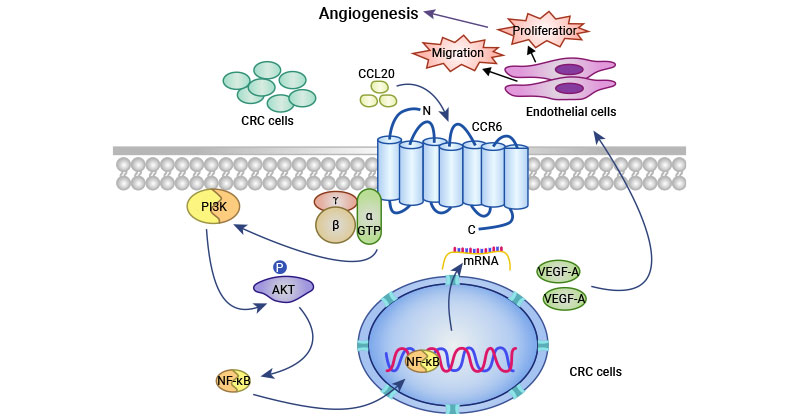
[16] Zhu, Cong-cong, et al. "CCR6 promotes tumor angiogenesis via the AKT/NF-κB/VEGF pathway in colorectal cancer." Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1864.2 (2018): 387-397.
[17] Bracke, Ken R., et al. "Cigarette smoke-induced pulmonary inflammation and emphysema are attenuated in CCR6-deficient mice." The Journal of Immunology 177.7 (2006): 4350-4359.
[18] Wan, Wuzhou, et al. "Genetic deletion of chemokine receptor Ccr6 decreases atherogenesis in ApoE-deficient mice." Circulation research 109.4 (2011): 374-381.
[19] Wan, Wuzhou, and Philip M. Murphy. "Regulation of atherogenesis by chemokine receptor CCR6." Trends in cardiovascular medicine 21.5 (2011): 140-144.
[20] Comerford, Iain, et al. "An immune paradox: how can the same chemokine axis regulate both immune tolerance and activation? CCR6/CCL20: a chemokine axis balancing immunological tolerance and inflammation in autoimmune disease." Bioessays 32.12 (2010): 1067-1076.
[21] Chung, So-Hyang, et al. "The CC chemokine receptor 6 (CCR6) is crucial for Th2-driven allergic conjunctivitis." Clinical Immunology 161.2 (2015): 110-119.
[22] Nandi, Bisweswar, et al. "Stromal CCR6 drives tumor growth in a murine transplantable colon cancer through recruitment of tumor-promoting macrophages." Oncoimmunology 5.8 (2016): e1189052.
[23] Ohtani, Haruo, Takashi Nakayama, and Osamu Yoshie. "In situ expression of the CCL20–CCR6 axis in lymphocyte‐rich gastric cancer and its potential role in the formation of lymphoid stroma." Pathology international 61.11 (2011): 645-651.
[24] Nandi, Bisweswar, et al. "CCR6, the sole receptor for the chemokine CCL20, promotes spontaneous intestinal tumorigenesis." PloS one 9.5 (2014): e97566.
[25] Kadomoto, Suguru, Kouji Izumi, and Atsushi Mizokami. "The CCL20-CCR6 axis in cancer progression." International journal of molecular sciences 21.15 (2020): 5186.
[26] Cook, Katherine W., et al. "CCL20/CCR6-mediated migration of regulatory T cells to the Helicobacter pylori-infected human gastric mucosa." Gut (2014): gutjnl-2013.
[27] Dellacasagrande, J., et al. "Liver metastasis of cancer facilitated by chemokine receptor CCR6." Scandinavian journal of immunology 57.6 (2003): 534-544.
[28] Rubie, Claudia, et al. "CCL20/CCR6 expression profile in pancreatic cancer." Journal of translational medicine 8 (2010): 1-8.
[29] Lian, Jingyao, et al. "Eomes promotes esophageal carcinoma progression by recruiting Treg cells through the CCL20‐CCR6 pathway." Cancer Science 112.1 (2021): 144-154.
[30] Zhang, X. G., et al. "CCR6 overexpression predicted advanced biological behaviors and poor prognosis in patients with gastric cancer." Clinical and Translational Oncology 18 (2016): 700-707.
[31] Xu, Lin, et al. "In situ prior proliferation of CD4+ CCR6+ regulatory T cells facilitated by TGF-β secreting DCs is crucial for their enrichment and suppression in tumor immunity." PLoS One 6.5 (2011): e20282.



Comments
Leave a Comment