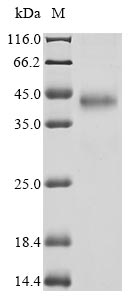
Recombinant Drosophila melanogaster GEO11329p1 (ITP) is produced in a mammalian cell expression system, which appears to ensure proper folding and post-translational modifications. The protein is expressed as the full length of its mature form, spanning amino acids 33 to 119, and carries a C-terminal hFc1 tag for easier detection and purification. The product achieves a purity of greater than 90%, as verified by SDS-PAGE analysis, making it suitable for various research applications.
GEO11329p1, also known as ITP, is a protein found in Drosophila melanogaster—the common fruit fly. It likely plays a crucial role in signaling pathways that are important for cellular communication and regulation. Scientists often study this protein for its involvement in key physiological processes, which has made it a significant focus in research related to insect biology and genetics.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
Based on the provided information, the recombinant ITP protein may have a high probability of correct folding due to the mammalian cell expression system, which can support proper eukaryotic folding and potential post-translational modifications. However, the C-terminal hFc1 tag (a large fusion tag, typically ~25 kDa) is attached to the mature ITP domain (33-119aa, ~10 kDa), which could sterically hinder the protein's native structure, especially if the C-terminal region is involved in receptor binding or function. ITP is a small peptide hormone, and the tag may disrupt its conformational integrity or activity. The purity >90% indicates low contaminants, but does not guarantee correct folding or bioactivity. Without experimental validation (e.g., circular dichroism for secondary structure, receptor binding assays, or functional tests in Drosophila systems), the protein's folding status and bioactivity remain uncertain. Therefore, while the expression system is favorable, the tag and lack of validation mean it cannot be assumed to be fully functional.
1. Ion Transport Peptide (ITP) Functional Studies in Drosophila Research
This recombinant ITP protein can be used for functional studies only if correct folding and bioactivity are experimentally verified. The mammalian cell expression system may support proper folding, but the C-terminal hFc1 tag could interfere with receptor binding or signaling. If misfolded, results from binding assays or pathway studies may be invalid. It is recommended to validate activity using in vitro assays (e.g., with Drosophila receptors) or in vivo complementation tests before concluding. The tag simplifies detection but may not preserve native function.
2. Antibody Development and Validation
This recombinant ITP can serve as an immunogen for antibody generation, but the hFc1 tag may dominate the immune response, leading to antibodies that primarily recognize the tag rather than ITP-specific epitopes. If correctly folded, antibodies might recognize native ITP; if misfolded, they could target non-conformational epitopes, reducing utility for detecting endogenous ITP. For reliable antibodies, validate using tag-free ITP or confirm specificity with Drosophila tissue samples. The high purity is beneficial but does not ensure antigen quality.
3. Protein-Protein Interaction Studies
The hFc1-tagged ITP can be used in pull-down assays, but interactions detected may be tag-mediated or non-specific if the protein is misfolded. The Fc tag enables efficient capture on protein A/G beads, but biologically relevant partners might not bind correctly without native conformation. Validate folding first and include controls (e.g., tag-only protein or untagged ITP) to minimize artifacts. Mass spectrometry results should be confirmed with orthogonal methods.
4. ELISA-Based Quantitative Assays
This recombinant ITP can be used as a standard in ELISA assays, but the hFc1 tag may affect antibody recognition if the tag blocks epitopes. For accurate quantification of endogenous ITP, ensure that antibodies used in the assay are specific to native ITP and not influenced by the tag. Validate the assay with spike-recovery experiments using Drosophila samples to confirm reliability. The high purity supports consistency but does not guarantee immunological accuracy.
Final Recommendation & Action Plan
To ensure reliable outcomes, first validate the folding and bioactivity of the recombinant ITP protein using techniques such as circular dichroism to assess secondary structure, surface plasmon resonance or competitive binding assays to confirm receptor interaction, and functional assays in Drosophila models (e.g., ion transport tests). If possible, remove the hFc1 tag via proteolytic cleavage and re-purify the tag-free protein for critical applications. For antibody development, use the current protein but characterize antibodies thoroughly against tag-free ITP and endogenous samples. In all studies, include appropriate controls, such as tag-only proteins or wild-type ITP standards, to account for potential artifacts. Prioritize validation before functional or quantitative applications to avoid misleading data.