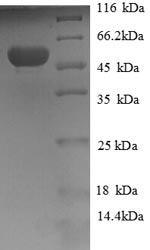
In e.coli cells, the generation of recombinant Human HAGH protein involves cloning a DNA fragment encoding the Human HAGH protein (50-308aa) into a plasmid vector, which is then transferred into the e.coli cells. Positive cells are selected, cultured, and induced to express the HAGH protein. A N-terminal GST tag is attached to the protein. Lysis of the cells allows for the harvest of the recombinant Human HAGH protein. The collected recombiant Human HAGH protein is subjected to affinity purification and is identified using SDS-PAGE and subsequent staining of the gel with Coomassie Brilliant Blue. Its purity is greater than 90%.