[1] Yu Xu, Jing Peng, Yizhong Yan, Min Gao, HongJing Zang, Lamei Cheng, Yu Zhou. (2025). CD19.
[2] Natalia M Sutherland, Baijun Zhou, Lingxiao Zhang, Mei-Sing Ong, Joseph S Hong, Andrew Pak, Katherine J Liu, Matthew J Frigault, Marcela V Maus, Joshua A Hill, Kerry Reynolds, Jolan E Walter, Carlos A Camargo, Sara Barmettler. (2025). Association of CD19.
[3] Katherine J Susa, Gary A Bradshaw, Robyn J Eisert, Charlotte M Schilling, Marian Kalocsay, Stephen C Blacklow, Andrew C Kruse. (2024). A spatiotemporal map of co-receptor signaling networks underlying B cell activation.
[4] Matthew T Witkowski, Soobeom Lee, Eric Wang, Anna K Lee, Alexis Talbot, Chao Ma, Nikolaos Tsopoulidis, Justin Brumbaugh, Yaqi Zhao, Kathryn G Roberts, Simon J Hogg, Sofia Nomikou, Yohana E Ghebrechristos, Palaniraja Thandapani, Charles G Mullighan, Konrad Hochedlinger, Weiqiang Chen, Omar Abdel-Wahab, Justin Eyquem, Iannis Aifantis. (2022). NUDT21 limits CD19 levels through alternative mRNA polyadenylation in B cell acute lymphoblastic leukemia.
[5] Yoshitaka Adachi, Seitaro Terakura, Masahide Osaki, Yusuke Okuno, Yoshitaka Sato, Ken Sagou, Yuki Takeuchi, Hirofumi Yokota, Kanae Imai, Peter Steinberger, Judith Leitner, Ryo Hanajiri, Makoto Murata, Hitoshi Kiyoi. (2024). Cullin-5 deficiency promotes chimeric antigen receptor T cell effector functions potentially via the modulation of JAK/STAT signaling pathway.
[6] Alberto J Millan, Vincent Allain, Indrani Nayak, Jeremy B Libang, Lilian M Quijada-Madrid, Janice S Arakawa-Hoyt, Gabriella Ureno, Allison Grace Rothrock, Avishai Shemesh, Oscar A Aguilar, Justin Eyquem, Jayajit Das, Lewis L Lanier. (2025). SYK negatively regulates ITAM-mediated human NK cell signaling and CD19-CAR NK cell efficacy.
[7] Marcelo C Pasquini, Zhen-Huan Hu, Kevin Curran, Theodore Laetsch, Frederick Locke, Rayne Rouce, Michael A Pulsipher, Christine L Phillips, Amy Keating, Matthew J Frigault, Dana Salzberg, Samantha Jaglowski, Joshua P Sasine, Joseph Rosenthal, Monalisa Ghosh, Daniel Landsburg, Steven Margossian, Paul L Martin, Manali K Kamdar, Peiman Hematti, Sarah Nikiforow, Cameron Turtle, Miguel-Angel Perales, Patricia Steinert, Mary M Horowitz, Amy Moskop, Lida Pacaud, Lan Yi, Raghav Chawla, Eric Bleickardt, Stephan Grupp. (2020). Real-world evidence of tisagenlecleucel for pediatric acute lymphoblastic leukemia and non-Hodgkin lymphoma.
[8] Yuekun Qi, Mingfeng Zhao, Yongxian Hu, Ying Wang, Ping Li, Jiang Cao, Ming Shi, Jiaqi Tan, Meng Zhang, Xia Xiao, Jieyun Xia, Sha Ma, Jianlin Qiao, Zhiling Yan, Hujun Li, Bin Pan, Wei Sang, Depeng Li, Zhenyu Li, Jianfeng Zhou, He Huang, Aibin Liang, Junnian Zheng, Kailin Xu. (2022). Efficacy and safety of CD19-specific CAR T cell-based therapy in B-cell acute lymphoblastic leukemia patients with CNSL.
[9] Ohad Benjamini, Avichai Shimoni, Michal J. Besser, Noga Shem‐Tov, Ivetta Danylesko, Ronit Yerushalmi, Drorit Merkel, Tamar Tadmor, David Lavie, Riva Fineman, Elad Jacobi, Arnon Nagler, Abraham Avigdor. (2020). Safety and Efficacy of CD19-CAR T Cells in Richter's Transformation after Targeted Therapy for Chronic Lymphocytic Leukemia.
[10] Rui Cui, Cuicui Lyu, Qing Li, Yanyu Jiang, Nan Mou, Zhenxing Yang, Xuxiang Liu, Qi Deng, Lanfang Li. (2020). Humanized anti‐CD19 chimeric antigen receptor‐T cell therapy is safe and effective in lymphoma and leukemia patients with chronic and resolved hepatitis B virus infection.
[11] John H Stone, Arezou Khosroshahi, Wen Zhang, Emanuel Della Torre, Kazuichi Okazaki, Yoshiya Tanaka, J Matthias Löhr, Nicolas Schleinitz, Lingli Dong, Hisanori Umehara, Marco Lanzillotta, Zachary S Wallace, Mikael Ebbo, George J Webster, Fernando Martinez Valle, Manu K Nayar, Cory A Perugino, Vinciane Rebours, Xinxin Dong, Yanping Wu, Qing Li, Nishi Rampal, Daniel Cimbora, Emma L Culver. (2025). Inebilizumab for Treatment of IgG4-Related Disease.
[12] Jeremias Motte, Melissa Sgodzai, Christiane Schneider-Gold, Nina Steckel, Thomas Mika, Tobias Hegelmaier, Dominic Borie, Aiden Haghikia, Dimitrios Mougiakakos, Roland Schroers, Ralf Gold. (2024). Treatment of concomitant myasthenia gravis and Lambert-Eaton myasthenic syndrome with autologous CD19-targeted CAR T cells.
[13] Artur Wilhelm, David Chambers, Fabian Müller, Aline Bozec, Ricardo Grieshaber-Bouyer, Thomas Winkler, Dimitrios Mougiakakos, Andreas Mackensen, Georg Schett, Gerhard Krönke. (2024). Selective CAR T cell-mediated B cell depletion suppresses IFN signature in SLE.
[14] Fabian Müller, Jule Taubmann, Laura Bucci, Artur Wilhelm, Christina Bergmann, Simon Völkl, Michael Aigner, Tobias Rothe, Ioanna Minopoulou, Carlo Tur, Johannes Knitza, Soraya Kharboutli, Sascha Kretschmann, Ingrid Vášová, Silvia Spoerl, Hannah Reimann, Luis E. Muñoz, Roman G. Gerlach, Simon Schäfer, Ricardo Grieshaber‐Bouyer, Anne‐Sophie Korganow, Dominique Farge, Dimitrios Mougiakakos, Aline Bözec, Thomas Winkler, Gerhard Krönke, Andréas Mackensen, Georg Schett. (2024). CD19 CAR T-Cell Therapy in Autoimmune Disease --- A Case Series with Follow-up.
[15] Janina Auth, Fabian Müller, Simon Völkl, Nadine Bayerl, Jörg H W Distler, Carlo Tur, Maria G Raimondo, Sara Chenguiti Fakhouri, Armin Atzinger, Birte Coppers, Markus Eckstein, Anna-Maria Liphardt, Tobias Bäuerle, Koray Tascilar, Michael Aigner, Sascha Kretschmann, Andreas Wirsching, Jule Taubmann, Melanie Hagen, Andrea-Hermina Györfi, Soraya Kharboutli, Tobias Krickau, Clara Dees, Silvia Spörl, Tobias Rothe, Thomas Harrer, Aline Bozec, Ricardo Grieshaber-Bouyer, Florian Fuchs, Torsten Kuwert, Carola Berking, Raymund E Horch, Michael Uder, Andreas Mackensen, Georg Schett, Christina Bergmann. (2025). CD19-targeting CAR T-cell therapy in patients with diffuse systemic sclerosis: a case series.
[16] Junli Wang, Wanyue Cao, Jinyan Huang, Yu Zhou, Rujia Zheng, Yu Lou, Jiaqi Yang, Jianghui Tang, Mao Ye, Zhengtao Hong, Jiangchao Wu, Haonan Ding, Yuquan Zhang, Jianpeng Sheng, Xinjiang Lu, Pinglong Xu, Xiongbin Lu, Xueli Bai, Tingbo Liang, Qi Zhang. (2025). Tumor-associated CD19+ macrophages induce immunosuppressive microenvironment in hepatocellular carcinoma.

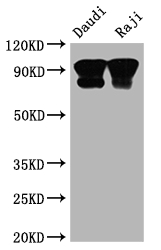
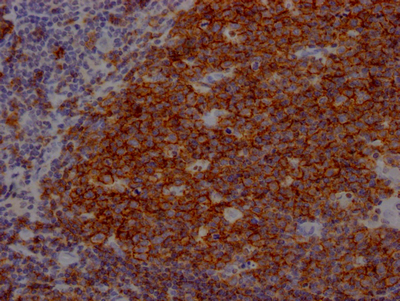
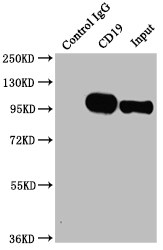
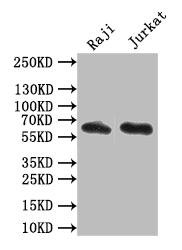


Comments
Leave a Comment