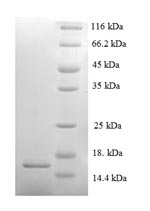
Amino acids 20-122 constitute the expression domain of recombinant Mouse Saa2. The calculated molecular weight for this Saa2 protein is 15.6 kDa. This protein is generated in a e.coli-based system. The Saa2 coding gene included the N-terminal 6xHis tag, which simplifies the detection and purification processes of the recombinant Saa2 protein in following stages of expression and purification.
The mouse serum amyloid A-2 protein (Saa2) is a major acute-phase protein primarily produced in the liver in response to inflammatory stimuli such as infection, tissue injury, or cytokine signaling. It belongs to the serum amyloid A (SAA) family of proteins, which are involved in various immune responses and inflammatory processes. Saa2 is implicated in modulating inflammation, lipid metabolism, and the acute-phase response. Its expression is tightly regulated and can be induced by pro-inflammatory cytokines like IL-6 and TNF-α. Research on Saa2 spans areas such as inflammation, immune responses, and metabolic disorders, aiming to elucidate its role in health and disease, including its potential as a biomarker or therapeutic target for inflammatory conditions.