HSPB1, commonly known as HSP27, is a small heat shock protein that plays a critical role in cellular stress responses, cytoskeletal organization, and apoptosis regulation. Phosphorylation at serine 78 represents a key regulatory modification that modulates HSPB1's chaperone activity and its interactions with client proteins. This phosphorylation event is particularly significant in signal transduction research, as it serves as a downstream readout for stress-activated kinase pathways and has been implicated in cancer progression, therapeutic resistance, and inflammatory responses.
This recombinant monoclonal antibody, clone 2D8, specifically recognizes the phosphorylated serine 78 residue of human HSPB1. Developed using recombinant technology in a rabbit host, this antibody offers the reproducibility and sequence-defined consistency that phospho-specific detection demands. Unlike traditional hybridoma-derived antibodies, recombinant production ensures that each lot performs identically, which is essential when tracking subtle changes in phosphorylation status across experimental conditions.
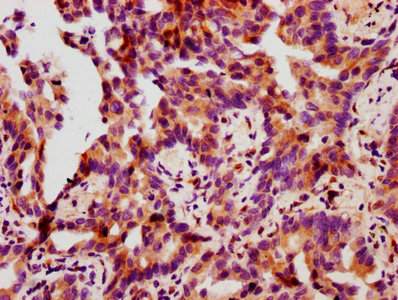
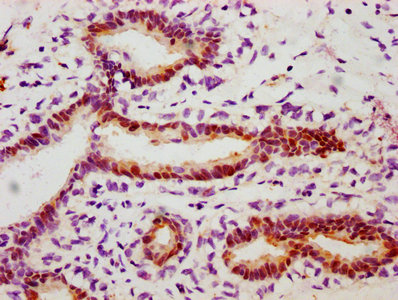
Validation studies demonstrate reliable performance across multiple applications. In western blot analysis, the antibody detects a band at the expected 27 kDa molecular weight in A549 lung adenocarcinoma cell lysates, with clear differential detection between EGF-treated and untreated conditions at working dilutions of 1:500 to 1:5000. For immunohistochemistry applications, the antibody has been validated in paraffin-embedded human lung cancer and breast cancer tissues at 1:50 to 1:200 dilutions using citrate buffer antigen retrieval, producing specific staining patterns suitable for tissue-based phosphorylation studies.
This phospho-specific antibody serves researchers investigating stress signaling, cancer biology, and therapeutic mechanisms where HSPB1 phosphorylation dynamics provide meaningful biological insights.