RNA polymerase II, the enzyme responsible for transcribing protein-coding genes into mRNA, undergoes dynamic phosphorylation at its C-terminal domain that serves as a critical regulatory mechanism for transcription. Phosphorylation at serine 2 of POLR2A marks the transition from transcription initiation to productive elongation, making this modification an essential marker for studying active transcription, gene regulation, and the coordination between RNA synthesis and co-transcriptional processing events.
This recombinant monoclonal antibody, clone 2G1, specifically recognizes the phosphorylated serine 2 residue of human POLR2A. Developed using recombinant technology in rabbit host, this antibody offers the consistency and reproducibility that phospho-specific detection demands. Each lot derives from a defined sequence, eliminating the variability that can compromise sensitive phosphorylation studies where signal specificity is paramount.
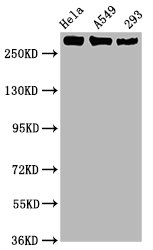
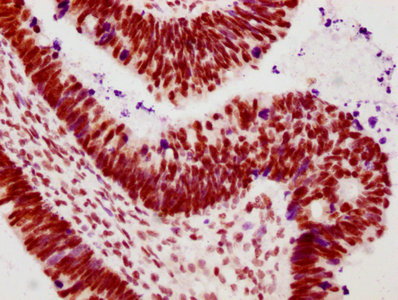
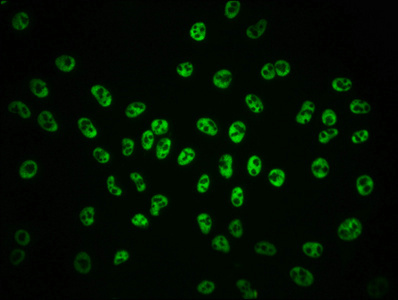
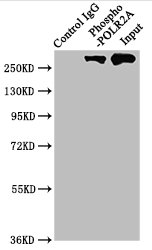
Validation across multiple applications demonstrates genuine experimental flexibility. Western blot analysis confirms detection of the expected 270 kDa band in HeLa, A549, and 293 cell lysates, with recommended dilutions ranging from 1:500 to 1:5000. Immunohistochemistry performed on paraffin-embedded human ovarian cancer tissue shows clear nuclear staining using citrate buffer antigen retrieval. Immunofluorescence in HeLa cells reveals the anticipated nuclear localization pattern consistent with active transcription sites. Immunoprecipitation has been validated in HeLa lysates, enabling enrichment studies and interaction analyses.
For researchers investigating transcriptional regulation, epigenetic mechanisms, or nuclear signaling pathways, this phospho-specific antibody provides a reliable tool for monitoring POLR2A serine 2 phosphorylation status across biochemical, imaging, and tissue-based experimental approaches.