CTSB (Cathepsin B) is a lysosomal cysteine protease that plays a role in protein degradation, tissue remodeling, and immune response. Recent research has highlighted its potential involvement in diseases such as Alzheimer's disease, cancer, COVID-19, and liver disease, and its potential as a therapeutic target in these contexts.
1. What is CTSB?
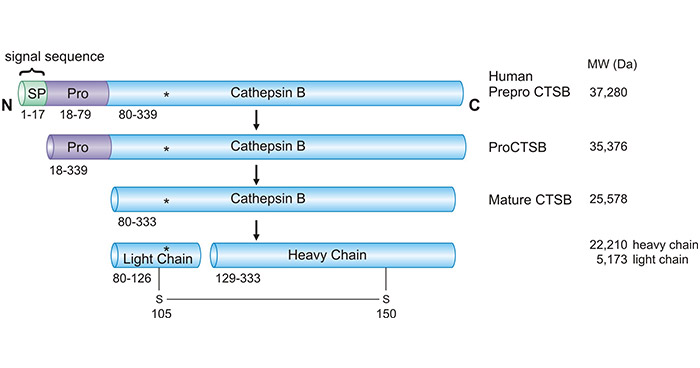
CTSB is a 36 kDa protein that is encoded by the CTSB gene. It contains a signal peptide, a propeptide, and a mature enzyme. CTSB is synthesized as a preproenzyme in the endoplasmic reticulum and then transported to lysosomes, where it is processed into its mature form upon cleavage of the N-terminal signal and pro-peptide regions by proteolytic cleavage.
Figure 1. CTSB Maturation: Inactive zymogen conversion to active CTSB
The mature CTSB protein is a single polypeptide chain that consists of 339 amino acids and has a pI of 5.1. It contains an active site cysteine residue responsible for its proteolytic activity, a binding site for its substrate, and several other conserved amino acid residues important for its structure and function.
CTSB is expressed in various tissues and cell types, including immune cells, neurons, and cancer cells. Its expression is regulated by multiple factors, including growth factors, cytokines, and transcription factors. CTSB mainly locates in secretory vesicles, cytoplasm, and nucleus, and also binds to the plasma membrane. CTSB is also secreted into the extracellular space, where it can participate in pathological processes such as tumor invasion and metastasis.
2. Functions of CTSB
The additional pH-sensitive occluding loop allows CTSB to act as an endopeptidase/exopeptidase depending upon pH. Therefore, CTSB has a broad substrate specificity and can degrade a variety of proteins, including collagen, elastin, and fibronectin, which is important for maintaining cellular homeostasis. It also has non-proteolytic functions, such as activating procathepsins and other enzymes.
CTSB plays an important role in multiple cellular activities, including tissue remodeling, antigen-presenting, autophagy, apoptosis, cell proliferation, hippocampal-dependent memory function, cell differentiation, and cell migration.
Dysregulation of CTSB has been linked to various diseases, including cancer, where it can promote tumor invasion and metastasis, and neurodegenerative disorders, where it can contribute to the accumulation of misfolded proteins in the brain. CTSB has also been implicated in cardiovascular disease, osteoporosis, and inflammatory disorders. Therefore, understanding the functions of CTSB is important for developing therapies to treat these conditions.
3. Latest Studies on CTSB
CTSB is a multi-functional protein involved in a wide range of physiological and pathological processes, making it a versatile and attractive target for research.
3.1 CTSB in Neurological Function
CTSB plays a crucial role in various neurological processes. Pedersen et al. revealed that CTSB is an exercise-induced muscle secretory factor associated with hippocampal functions [1]. It is also believed to have a role in the clearance of misfolded proteins in neurodegenerative diseases such as Alzheimer's disease [2]. Furthermore, CTSB has been linked to autophagy [3][4], a cellular process that helps to maintain neuronal homeostasis.
3.2 CTSB in cancer
The research conducted by Kaiming Ma and colleagues revealed a strong correlation between CTSB and malignant pathological subtypes, poorer prognosis, as well as immune cell infiltration and immunosuppression in gliomas [5]. Furthermore, CTSB appears to play a significant role in signal transduction, immune and inflammatory response, modulation of the immune response, leukocyte migration, interferon-gamma-mediated signaling pathway, and antigen processing and presentation in gliomas. Considering these findings, CTSB holds promise as both a biomarker and target in the diagnosis, treatment and prognostic assessment of gliomas.
A multitude of cancers, including but not limited to pancreatic cancer, gastric cancer, colon cancer, breast cancer, acute myelogenous leukemia, oral squamous cell carcinoma, and thyroid cancer, have identified CTSB as a significant biomarker and a potential therapeutic target. CSTB upregulation has been detected in hepatocellular carcinoma and ovarian clear cell carcinoma [6][7], while in gastric cancer cells, CSTB knockdown resulted in reduced proliferation and migration [8].
3.3 CTSB in COVID-19
Studies have demonstrated that CTSB is involved in the entry of SARS-CoV-2, the virus that causes COVID-19, into host cells [9][10]. Inhibition of CTSB activity has been suggested as a potential therapeutic strategy for COVID-19.
Furthermore, research has indicated that increased CTSB expression is linked to the severity of COVID-19. In severe cases of the disease, the level of CTSB in the blood is higher, suggesting that CTSB may contribute to the immune response to the virus. It has been suggested that CTSB may play a role in the cytokine storm, an excessive immune response that damages tissues and organs and can lead to death.
These findings highlight the potential of CTSB as a therapeutic target for the treatment of COVID-19. Researchers are investigating drugs that can inhibit the activity of CTSB, potentially reducing the virus's ability to infect cells and prevent the cytokine storm. Several drugs, such as camostat mesylate and nafamostat, have shown promise in inhibiting CTSB activity in preclinical studies and clinical trials.
3.4 CTSB in Liver Disease
Wenqian Fang et al. found that CTSB deficiency improved liver functions (such as liver lipid deposition and inflammatory cell infiltration ) in mice with non-alcoholic steatohepatitis (NASH) by inhibiting de novo lipogenesis and liver inflammation and fibrosis [11].
4. CTSB-related Products
CUSABIO provides some CTSB products for scientific research, including CTSB recombinant proteins, CTSB antibodies, and CTSB ELISA kits.
Overall, CTSB is a protein with diverse functions and implications in various disease processes. Its broad substrate specificity, non-proteolytic functions, and involvement in many cellular processes make it a challenging target for drug development. Recent advances in understanding the structure and function of CTSB have led to the development of several potent and selective CTSB inhibitors. These inhibitors may have therapeutic potential in treating diseases such as cancer, Alzheimer's, COVID-19, and liver disease. Further research is needed to fully understand the mechanisms of CTSB action and to optimize the development of CTSB-targeted therapeutics.
References
[1] Pedersen, B. K. (2019). Physical activity and muscle–brain crosstalk [J]. Nat. Rev. Endocrinol. 15, 383–392.
[2] Hook, V., Yoon, M., et al. (2020). Cathepsin B in Neurodegeneration of Alzheimer’s Disease, Traumatic Brain Injury, and Related Brain Disorders [J]. Biochimica et biophysica acta. Proteins and proteomics, 1868(8), 140428.
[3] Iwama, H., Mehanna, S., Imasaka, M. et al. Cathepsin B and D deficiency in the mouse pancreas induces impaired autophagy and chronic pancreatitis [J]. Sci Rep 11, 6596 (2021).
[4] Yongsheng Jiang, Lijie Han, et al. Cystatin B increases autophagic flux by sustaining proteolytic activity of cathepsin B and fuels glycolysis in pancreatic cancer: CSTB orchestrates autophagy and glycolysis in PDAC [J]. Volume12, Issue12, December 2022, e1126.
[5] Ma, K., Chen, X., Liu, W. et al. CTSB is a negative prognostic biomarker and therapeutic target associated with immune cells infiltration and immunosuppression in gliomas [J]. Sci Rep 12, 4295 (2022).
[6] Takaya A, Peng WX, Ishino K, et al. Cystatin B as a potential diagnostic biomarker in ovarian clear cell carcinoma [J]. Int J Oncol. 2015;46(4):1573-1581.
[7] Lin YY, Chen ZW, Lin ZP, et al. Tissue levels of Stefin A and Stefin B in hepatocellular carcinoma [J]. Anat Rec. 2016;299(4):428-438.
[8] Zhang J, Shi Z, Huang J, Zou X. CSTB downregulation promotes cell proliferation and migration and suppresses apoptosis in gastric cancer SGC-7901 cell line [J]. Oncol Res. 2016;24(6):487-494.
[9] Bollavaram, K., Leeman, T. H., et al. (2021). Multiple Sites on SARS‐CoV ‐2 Spike Protein Are Susceptible to Proteolysis by Cathepsins B, K, L, S, and V [J]. Protein Sci. 30 (6), 1131–1143.
[10] Ou X, Liu Y, Lei X, et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV [J]. Nat Commun. 2020 Mar 27;11(1):1620.
[11] Fang, W., Deng, Z., et al. (2020). Cathepsin B deficiency ameliorates liver lipid deposition, inflammatory cell infiltration, and fibrosis after diet-induced nonalcoholic steatohepatitis [J]. Translational research : The journal of laboratory and clinical medicine, 222, 28.
CUSABIO team. Latest Studies on CTSB — A Multi-functional Protein. https://www.cusabio.com/c-19849.html



Comments
Leave a Comment