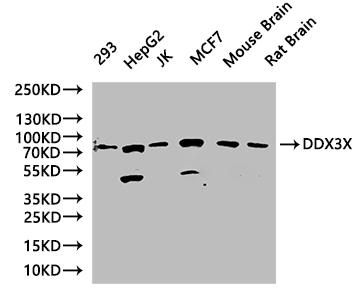
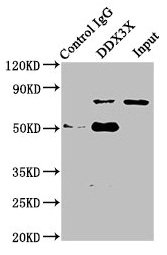
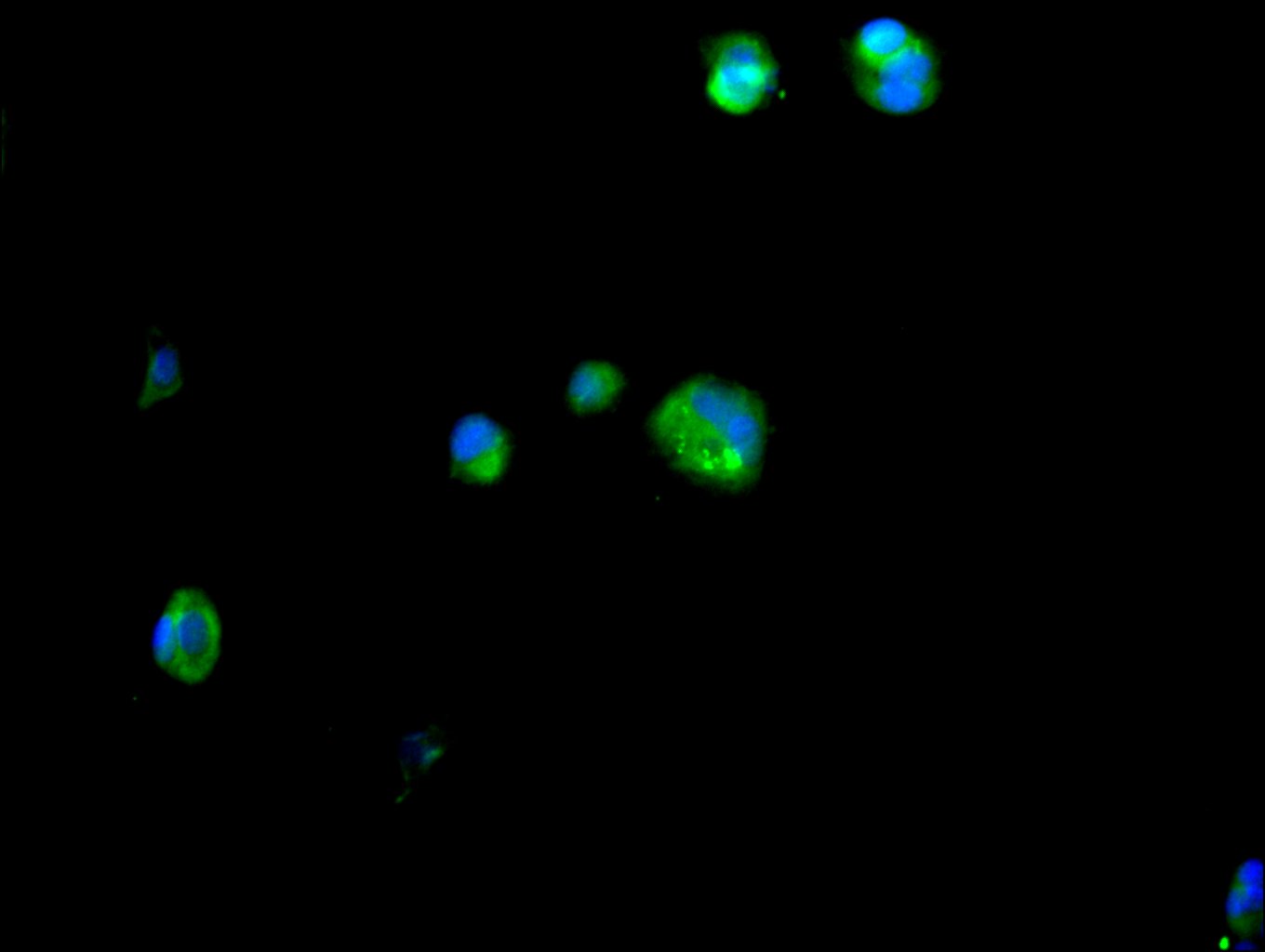
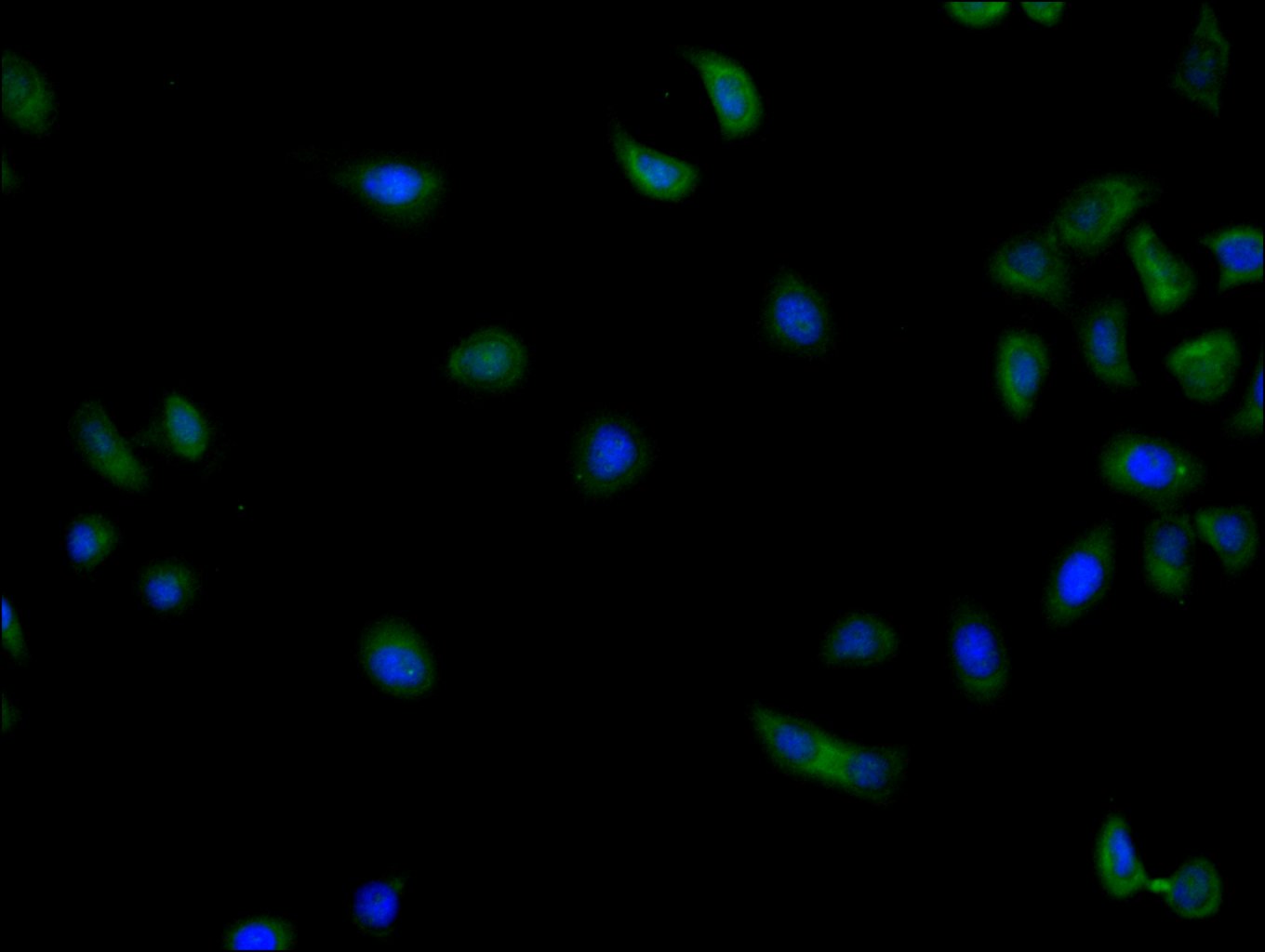
Multifunctional ATP-dependent RNA helicase. The ATPase activity can be stimulated by various ribo-and deoxynucleic acids indicative for a relaxed substrate specificity. In vitro can unwind partially double-stranded DNA with a preference for 5'-single-stranded DNA overhangs. Binds RNA G-quadruplex (rG4s) structures, including those located in the 5'-UTR of NRAS mRNA. Involved in many cellular processes, which do not necessarily require its ATPase/helicase catalytic activities. Involved in transcription regulation. Positively regulates CDKN1A/WAF1/CIP1 transcription in an SP1-dependent manner, hence inhibits cell growth. This function requires its ATPase, but not helicase activity. CDKN1A up-regulation may be cell-type specific. Binds CDH1/E-cadherin promoter and represses its transcription. Potentiates HNF4A-mediated MTTP transcriptional activation; this function requires ATPase, but not helicase activity. Facilitates HNF4A acetylation, possibly catalyzed by CREBBP/EP300, thereby increasing the DNA-binding affinity of HNF4 to its response element. In addition, disrupts the interaction between HNF4 and SHP that forms inactive heterodimers and enhances the formation of active HNF4 homodimers. By promoting HNF4A-induced MTTP expression, may play a role in lipid homeostasis. May positively regulate TP53 transcription. Associates with mRNPs, predominantly with spliced mRNAs carrying an exon junction complex (EJC). Involved in the regulation of translation initiation. Not involved in the general process of translation, but promotes efficient translation of selected complex mRNAs, containing highly structured 5'-untranslated regions (UTR). This function depends on helicase activity. Might facilitate translation by resolving secondary structures of 5'-UTRs during ribosome scanning. Alternatively, may act prior to 43S ribosomal scanning and promote 43S pre-initiation complex entry to mRNAs exhibiting specific RNA motifs, by performing local remodeling of transcript structures located close to the cap moiety. Independently of its ATPase activity, promotes the assembly of functional 80S ribosomes and disassembles from ribosomes prior to the translation elongation process. Positively regulates the translation of cyclin E1/CCNE1 mRNA and consequently promotes G1/S-phase transition during the cell cycle. May activate TP53 translation. Required for endoplasmic reticulum stress-induced ATF4 mRNA translation. Independently of its ATPase/helicase activity, enhances IRES-mediated translation; this activity requires interaction with EIF4E. Independently of its ATPase/helicase activity, has also been shown specifically repress cap-dependent translation, possibly by acting on translation initiation factor EIF4E. Involved in innate immunity, acting as a viral RNA sensor. Binds viral RNAs and promotes the production of type I interferon (IFN-alpha and IFN-beta). Potentiate MAVS/DDX58-mediated induction of IFNB in early stages of infection. Enhances IFNB1 expression via IRF3/IRF7 pathway and participates in NFKB activation in the presence of MAVS and TBK1. Involved in TBK1 and IKBKE-dependent IRF3 activation leading to IFNB induction, acts as a scaffolding adapter that links IKBKE and IRF3 and coordinates their activation. Involved in the TLR7/TLR8 signaling pathway leading to type I interferon induction, including IFNA4 production. In this context, acts as an upstream regulator of IRF7 activation by MAP3K14/NIK and CHUK/IKKA. Stimulates CHUK autophosphorylation and activation following physiological activation of the TLR7 and TLR8 pathways, leading to MAP3K14/CHUK-mediated activatory phosphorylation of IRF7. Also stimulates MAP3K14/CHUK-dependent NF-kappa-B signaling. Negatively regulates TNF-induced IL6 and IL8 expression, via the NF-kappa-B pathway. May act by interacting with RELA/p65 and trapping it in the cytoplasm. May also bind IFNB promoter; the function is independent of IRF3. Involved in both stress and inflammatory responses. Independently of its ATPase/helicase activity, required for efficient stress granule assembly through its interaction with EIF4E, hence promotes survival in stressed cells. Independently of its helicase activity, regulates NLRP3 inflammasome assembly through interaction with NLRP3 and hence promotes cell death by pyroptosis during inflammation. This function is independent of helicase activity. Therefore DDX3X availability may be used to interpret stress signals and choose between pro-survival stress granules and pyroptotic NLRP3 inflammasomes and serve as a live-or-die checkpoint in stressed cells. In association with GSK3A/B, negatively regulates extrinsic apoptotic signaling pathway via death domain receptors, including TNFRSF10B, slowing down the rate of CASP3 activation following death receptor stimulation. Cleavage by caspases may inactivate DDX3X and relieve the inhibition. Independently of its ATPase/helicase activity, allosteric activator of CSNK1E. Stimulates CSNK1E-mediated phosphorylation of DVL2, thereby involved in the positive regulation of Wnt/beta-catenin signaling pathway. Also activates CSNK1A1 and CSNK1D in vitro, but it is uncertain if these targets are physiologically relevant. ATPase and casein kinase-activating functions are mutually exclusive. May be involved in mitotic chromosome segregation.; (Microbial infection) Facilitates hepatitis C virus (HCV) replication. During infection, HCV core protein inhibits the interaction between MAVS and DDX3X and therefore impairs MAVS-dependent INFB induction and might recruit DDX3X to HCV replication complex.; (Microbial infection) Facilitates HIV-1 replication. Acts as a cofactor for XPO1-mediated nuclear export of HIV-1 Rev RNAs. This function is strongly stimulated in the presence of TBK1 and requires DDX3X ATPase activity.; (Microbial infection) Facilitates Zika virus (ZIKV) replication.; (Microbial infection) Facilitates Dengue virus (DENV) replication.; (Microbial infection) Facilitates Venezuelan equine encephalitis virus (VEEV) replication.