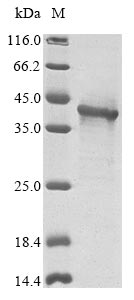
This recombinant Human IFNK protein is typically achieved by the manipulation of IFNK gene expression in E.coli cells so that it expresses large amounts of a recombinant IFNK gene. In order to get enough amount of the IFNK protein, strain selection, codon optimization, fusion systems, co-expression, mutagenesis, and isotope labeling techniques are used. Finally, the IFNK protein is isolated from the samples such as cell lysates or medium. Protein refolding, cleavage of fusion moieties and chromatography techniques are involved in the protein purification process.
IFNK is a multifunctional type I IFN that elicits autoimmunity when transgenically expressed in the β cells of the pancreatic islets in mice. IFNK is induced after viral infection or treatment of cells with double-stranded RNA (dsRNA). IFNK signaling stimulates a collection of genes, including IRF1, STAT1, MXA, PKR, and OAS1, which are common to signaling via the type I interferon receptor, and modulates the release of cytokines from monocytes. Different from other members of the type I IFN family, IFNK is constitutively and highly expressed in human keratinocytes. IFNK plays an important role in keratinocyte host defense against herpes simplex virus-1 (HSV-1).