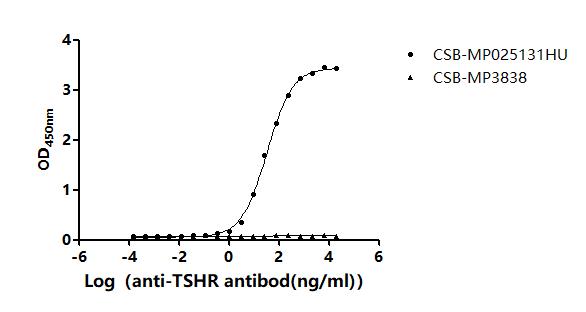
Thyrotropin receptor plays a central role in thyroid hormone regulation and is a primary autoantigen in Graves' disease, making functional receptor preparations critical for autoimmune research and therapeutic antibody development. This full-length mature protein (aa 21–764) is presented as virus-like particles that preserve native receptor conformation, enabling physiologically relevant binding interactions. Functional ELISA validation demonstrates specific antibody binding with an EC50 of 30.81–36.12 ng/mL when the receptor is immobilized at 10 μg/mL, confirming that the VLP format maintains epitope accessibility suitable for antibody screening, epitope mapping studies, and immunoassay development. The preparation meets endotoxin specifications below 1.0 EU/μg, aligning with standards expected in cell-based functional assays where low endotoxin levels are essential to avoid confounding inflammatory responses that could obscure receptor-mediated signaling events.