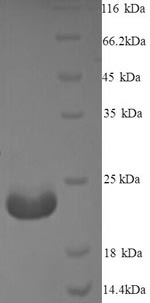
This recombinant Mycobacterium tuberculosis Immunogenic protein MPT64 gets expressed in a yeast system and covers the full length of the mature protein from amino acids 24 to 228. An N-terminal 6xHis-tag is included to make purification and detection more straightforward. SDS-PAGE analysis confirms the protein shows purity greater than 90%, which should provide reliable performance in research applications.
MPT64 appears to be a well-characterized immunogenic protein from Mycobacterium tuberculosis that researchers have studied extensively for its role in the immune response to tuberculosis. It represents a significant component in understanding how hosts and pathogens interact and seems crucial for developing diagnostic tools and vaccines. The research community has found that MPT64 helps clarify tuberculosis pathogenesis and immunity, though much remains to be discovered.
Potential Applications
Note: The applications listed below are based on what we know about this protein's biological functions, published research, and experience from experts in the field. However, we haven't fully tested all of these applications ourselves yet. We'd recommend running some preliminary tests first to make sure they work for your specific research goals.
Mycobacterium tuberculosis MPT64 is an immunogenic protein that requires proper folding for its antigenic properties and potential functional roles. The yeast expression system provides a eukaryotic environment that supports proper folding and some post-translational modifications, increasing the probability of correct folding compared to bacterial systems. The N-terminal 6xHis tag is relatively small and unlikely to significantly interfere with the protein's antigenic epitopes. While the protein may be correctly folded, experimental validation is recommended to confirm its structural integrity and antigenic activity.
1. Antibody Development and Characterization
This application is highly suitable regardless of folding status. Antibody development relies primarily on antigenic sequence recognition. If correctly folded (verified), the protein excels for generating conformation-sensitive antibodies that recognize native MPT64 epitopes. If misfolded/unverified, it remains suitable for producing antibodies against linear epitopes, which are still valuable for diagnostic and research applications.
2. ELISA-Based Binding Assays
This application is well-suited for immunological detection. If correctly folded (verified), the protein serves as an excellent antigen for detecting anti-MPT64 antibodies in serological assays. If misfolded/unverified, it remains effective as a coating antigen for ELISA since antibody binding primarily depends on linear epitopes.
3. Immunoblotting and Protein Detection Studies
This application is highly reliable. The recombinant protein works excellently as a positive control in Western blot experiments regardless of folding status. The known molecular weight and His-tag facilitate accurate identification and molecular weight standardization.
4. Affinity Purification and Pull-Down Experiments
This application requires folding validation. If correctly folded (verified), the protein can identify physiological interaction partners involved in M. tuberculosis pathogenesis. If misfolded/unverified, there is a high risk of non-specific binding or failure to identify genuine interactions.
Final Recommendation & Action Plan
The yeast expression system provides favorable conditions for producing correctly folded MPT64 protein, making it highly suitable for immunological applications. Begin with Applications 1, 2, and 3 for antibody development, ELISA assays, and immunoblotting controls, as these are reliable regardless of folding status. For Application 4 (interaction studies), first validate protein folding through biophysical characterization (e.g., circular dichroism spectroscopy, size-exclusion chromatography). Once correct folding is verified, proceed with pull-down experiments to identify genuine interaction partners. If misfolding is detected, limit the protein's use to immunological applications only.