| Drug |
Drug Type |
Indications Under Investigation (Disease Names) |
Research Institutions |
Highest Development Stage |
| Nivolumab/Relatlimab |
Monoclonal Antibody |
Metastatic melanoma, unresectable melanoma, melanoma, tumor, metastatic colorectal cancer, metastatic microsatellite-stable colorectal cancer, etc. |
Bristol Myers Squibb Co., Bristol-Myers Squibb Australia Pty Ltd., Bristol-Myers Squibb Pharma EEIG, Bristol-Myers Squibb (China) Investment Co., Ltd., Ono Pharmaceutical Co., Ltd., Shanghai Bristol-Myers Squibb Pharmaceuticals Co., Ltd. |
Approved |
| Fianlimab/Cemiplimab |
Monoclonal Antibody |
Unresectable melanoma, head and neck squamous cell carcinoma |
Regeneron Pharmaceuticals, Inc. |
Phase III |
| Recombinant Human Lymphocyte Activation Gene-3 (hLAG-3) Fusion Protein (Immutep)
|
Fc Fusion Protein |
Metastatic non-small cell lung cancer, metastatic HER2-negative breast cancer, HER2-positive metastatic breast cancer, soft tissue sarcoma, advanced head and neck squamous cell carcinoma, breast cancer, non-small cell lung cancer, head and neck squamous cell carcinoma, etc. |
Immutep SAS, Taizhou Yibai Kang Pharmaceutical Technology Co., Ltd., Merck Sharp & Dohme LLC, Immutep Ltd., Taizhou Yiteng Jingang Pharmaceutical Co., Ltd. |
Phase III |
| Alcestobart |
Monoclonal Antibody |
Tumor, metastatic esophageal squamous cell carcinoma, esophageal squamous cell carcinoma, solid tumor, colorectal cancer, nasopharyngeal cancer, microsatellite-stable colorectal cancer, advanced cancer, advanced malignant solid tumor, etc. |
Guangzhou BeiGene Biopharmaceutical Co., Ltd., Boqian Biotech Co., Ltd., BeiGene (Guangzhou) Pharmaceutical Co., Ltd., BeOne Medicines Ltd., Nanjing Weiling Bio-Technology Co., Ltd. |
Phase III |
| Relatlimab |
Monoclonal Antibody |
Recurrent non-squamous non-small cell lung cancer, unresectable melanoma, renal cell carcinoma, hepatocellular carcinoma, metastatic non-small cell lung cancer, etc. |
Bristol Myers Squibb Co., Shanghai Bristol-Myers Squibb Pharmaceuticals Co., Ltd., Ono Pharmaceutical Co., Ltd. |
Phase III |
| Favezelimab/Pembrolizumab |
Monoclonal Antibody |
Colorectal adenocarcinoma, metastatic colorectal cancer, cutaneous squamous cell carcinoma, mismatch repair-deficient endometrial cancer, refractory classical Hodgkin lymphoma, etc. |
Merck Sharp & Dohme Research (China) Co., Ltd., Merck Sharp & Dohme Corp., Merck Sharp & Dohme LLC, Merck & Co., Inc. |
Phase III |
| Fianlimab |
Monoclonal Antibody |
Melanoma, metastatic melanoma, advanced non-small cell lung cancer, locally advanced non-small cell lung cancer, muscle-invasive bladder cancer, etc. |
Regeneron Pharmaceuticals, Inc., Memorial Sloan Kettering Cancer Center, Sanofi |
Phase III |
| Tepotinib |
Bispecific Antibody |
Gastroesophageal junction cancer, HER2-positive gastric cancer, metastatic HER2-positive gastroesophageal junction cancer |
MacroGenics, Inc. |
Phase II/III |
| Nivolumab/Relatlimab/Hyaluronidase |
Monoclonal Antibody |
Metastatic melanoma |
Bristol Myers Squibb Co. |
Phase II |
| FS-118 |
Bispecific Antibody |
Non-small cell lung cancer, diffuse large B cell lymphoma, head and neck tumor, hematologic tumor, colorectal cancer |
Merck Serono SA, F-star Therapeutics, Inc., invoX Pharma Ltd., F-Star Delta Ltd., F Star Co., Ltd. |
Phase II |
| GSK-2831781 |
Monoclonal Antibody |
Ulcerative colitis, psoriasis |
Immutep Ltd. |
Phase II |
| Tuparstobart |
Monoclonal Antibody |
Metastatic Merkel cell carcinoma, head and neck squamous cell carcinoma, metastatic melanoma, unresectable melanoma |
Incyte Biosciences International SARL, Incyte Corp. |
Phase II |
| HLX-26 |
Monoclonal Antibody |
Advanced pulmonary non-squamous non-small cell lung cancer, advanced non-small cell lung cancer, metastatic non-small cell lung cancer, lymphoma, solid tumor, metastatic solid tumor, advanced malignant solid tumor, hepatocellular carcinoma |
Shanghai Henlius Biotechnology Co., Ltd., Shanghai Fosun Pharmaceutical Industrial Development Co., Ltd., Shanghai Henlius Biopharmaceutical Co., Ltd. |
Phase II |
| DNV-3 |
Monoclonal Antibody |
Melanoma, esophageal squamous cell carcinoma, gastroesophageal junction cancer, hepatocellular carcinoma, lymphoma, small cell lung cancer, etc. |
Zhejiang Timai Pharmaceutical Co., Ltd. |
Phase II |
| Tombestomig |
Bispecific Antibody |
PD-L1-positive triple-negative breast cancer, renal cell carcinoma, locally advanced renal cell carcinoma, locally advanced urothelial carcinoma, urothelial carcinoma, etc. |
Hoffmann-La Roche, Inc., Roche Holding AG, F. Hoffmann-La Roche Ltd., Roche (China) Investment Co., Ltd. |
Phase II |
| Miptenalimab |
Monoclonal Antibody |
Esophageal adenocarcinoma, advanced cancer, advanced endometrial cancer, locally advanced unresectable gastric adenocarcinoma, melanoma, etc. |
Boehringer Ingelheim GmbH |
Phase II |
| Cugrastomig |
Bispecific Antibody |
Colorectal adenocarcinoma, non-small cell lung cancer, non-small cell lung cancer stage IIIA, head and neck squamous cell carcinoma, etc., liver metastasis, melanoma, unresectable melanoma, refractory classical Hodgkin lymphoma |
Zhongshan Kangfang Biomedical Co., Ltd., Kangfang Huike (Shanghai) Biotechnology Co., Ltd. |
Phase I/II |
| Fanastomig |
Bispecific Antibody |
Solid tumor |
Anmab Biotechnology (Suzhou) Co., Ltd. |
Phase I/II |
| Recombinant Human Anti-LAG-3 Monoclonal Antibody (Guangzhou Yuheng Biotechnology/Wuxi AppTec) |
Monoclonal Antibody |
Advanced non-small cell lung cancer, advanced malignant solid tumor, melanoma |
Guangzhou Yuheng Biotechnology Co., Ltd. |
Phase I/II |
| Favezelimab |
Monoclonal Antibody |
Hematologic tumor |
Merck Sharp & Dohme LLC |
Phase I/II |
| TQB-2223/Penpulimab |
Monoclonal Antibody |
Advanced cancer |
Zhengda Tianqing Pharmaceutical Group Co., Ltd. |
Phase I |
| AK137 |
Bispecific Antibody |
Advanced cancer |
Zhongshan Kangfang Biomedical Co., Ltd. |
Phase I |
| INCA32459 |
Bispecific Antibody |
Metastatic head and neck squamous cell carcinoma, unresectable melanoma |
Incyte Corp. |
Phase I |
| IBI-323 |
Bispecific Antibody |
Advanced cancer |
Innovent Biologics (Suzhou) Co., Ltd. |
Phase I |
| IMP-761 |
Monoclonal Antibody |
Autoimmune diseases |
Immutep Ltd. |
Phase I |
| ABL-501 |
Bispecific Antibody |
Advanced malignant solid tumor |
ABL Bio, Inc. |
Phase I |
| Negalstobart |
Monoclonal Antibody |
Diffuse large B cell lymphoma, advanced cancer, gastroesophageal junction cancer, squamous non-small cell lung cancer, nasopharyngeal cancer |
Innovent Biologics (Suzhou) Co., Ltd. |
Phase I |
| TQB-2223 |
Monoclonal Antibody |
Advanced hepatocellular carcinoma, advanced cancer, cholangiocarcinoma, ampullary cancer of Vater, gallbladder cancer, recurrent solid tumor, tumor metastasis |
Symphogen A/S, Mingrui Pharmaceutical Co., Ltd., Zhengda Tianqing Pharmaceutical Group Co., Ltd., Clarusun Pharmaceutical Corp. |
Phase I |
| ZGGS15 |
Bispecific Antibody |
Advanced malignant solid tumor, solid tumor |
Suzhou Zelgen Biopharmaceuticals Co., Ltd. |
Phase I |
| Recombinant Humanized Monoclonal Antibody MIL98 |
Monoclonal Antibody |
Advanced lymphoma, melanoma |
Beijing Tiantan Bioscience Co., Ltd. |
Phase I |
| BJ-007 |
Monoclonal Antibody |
Infection |
Shanghai BioJie Pharmaceutical Co., Ltd. |
Clinical Application Approval |

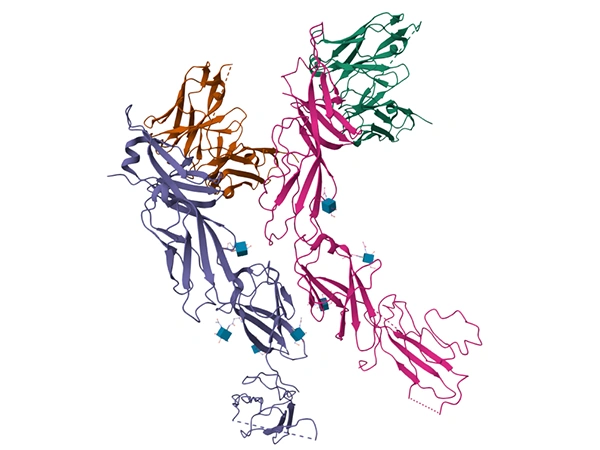
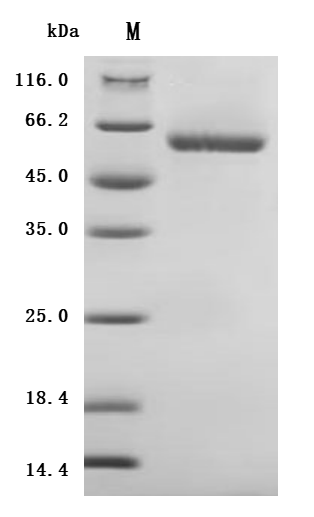
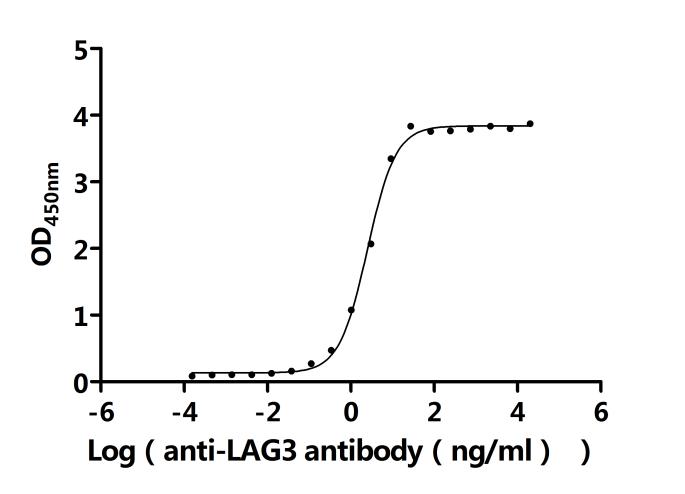
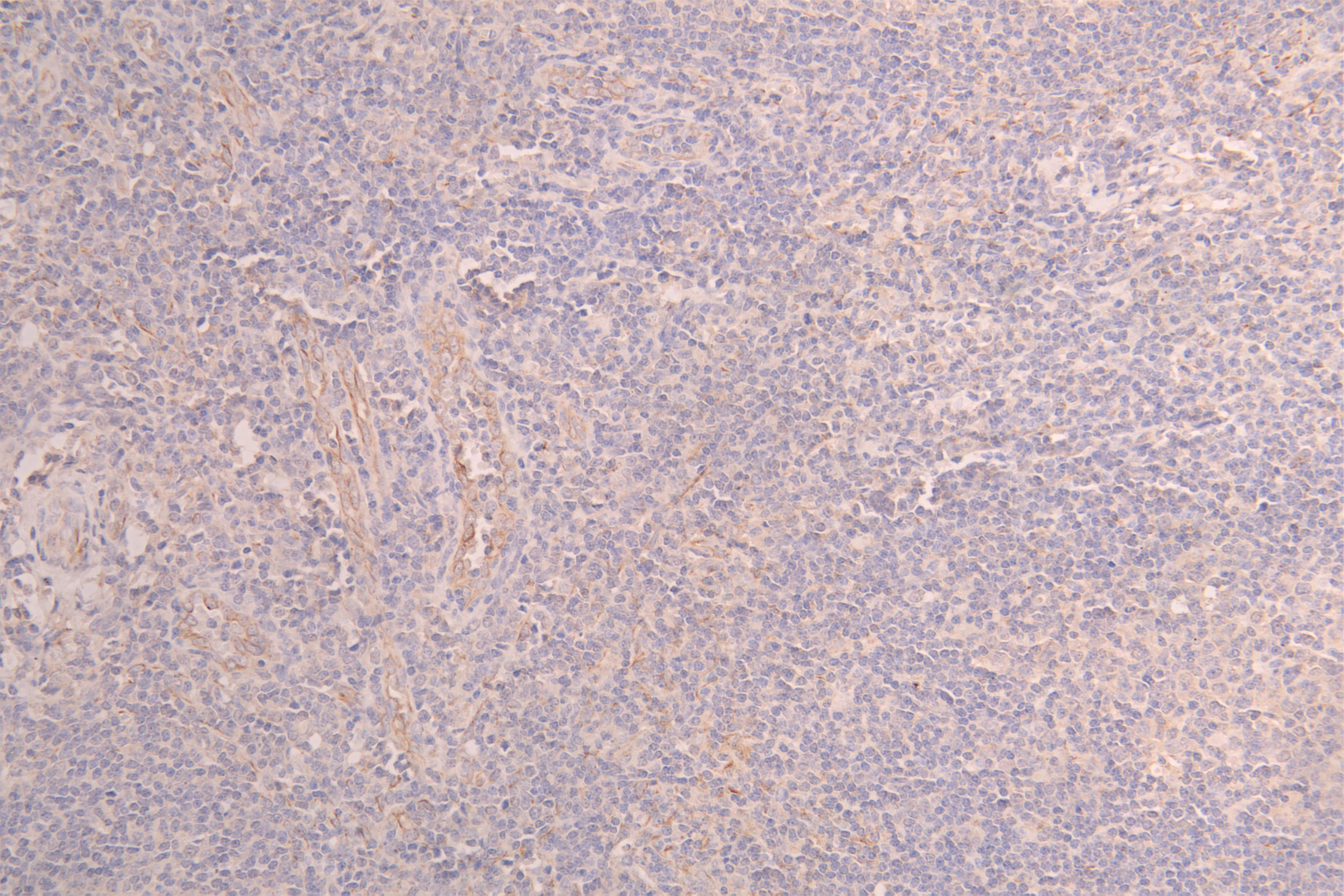
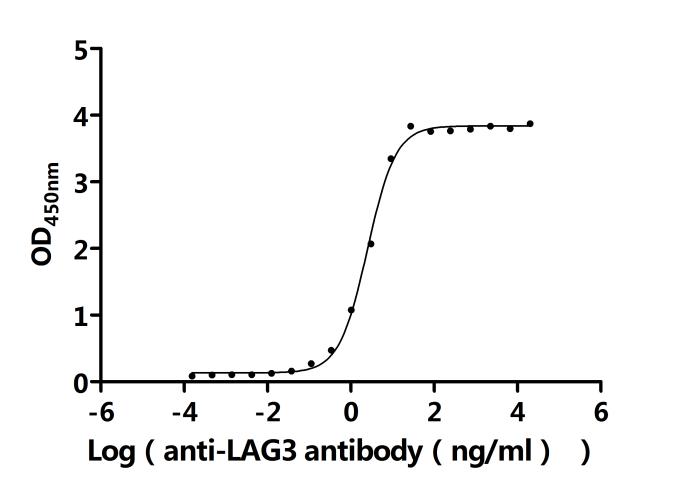
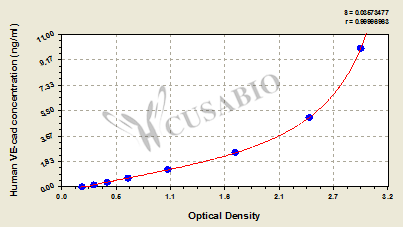
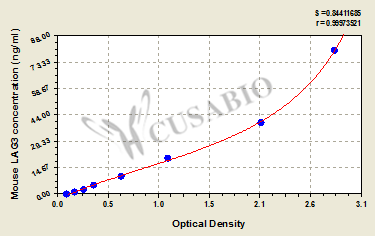


Comments
Leave a Comment