Brain-derived neurotrophic factor (BDNF) is the most widely studied core member of the neurotrophin family, playing an irreplaceable role in central nervous system development, neuronal survival, and synaptic plasticity regulation. BDNF activates downstream signaling pathways such as PI3K/Akt and MAPK/ERK through the TrkB receptor, regulating neuronal survival, axon growth, and cognitive function. Abnormal BDNF expression is closely associated with various neuropsychiatric diseases including Alzheimer's disease, Parkinson's disease, depression, and schizophrenia, while also having important functions in pain regulation, drug dependence, and nerve injury repair. This review systematically summarizes the gene structure, protein processing, signaling pathways, disease associations, and targeted therapeutic progress of BDNF, providing a reference framework for related research and clinical translation.
1. Biological Characteristics and Signaling Pathways of BDNF
1.1 BDNF Gene Structure and Protein Processing
The BDNF gene has a complex transcriptional regulatory structure, containing multiple promoters and exons. Transcription driven by different promoters produces multiple mRNA isoforms, which are basically identical in the coding region but differ in the 5' untranslated region, thus conferring different expression regulatory characteristics [1]. Neuronal activity regulates BDNF expression through Ca²⁺ signaling and transcription factors such as cAMP response element-binding protein (CREB), but this regulation differs significantly between brain regions [2].
In addition to neuronal activity regulation, various hormones and transcription factors are also involved in BDNF expression regulation. For example, estradiol, glucocorticoids, and myocyte enhancer factor-2 (MEF2) can all affect BDNF transcription levels by acting on the BDNF promoter region [3]. Furthermore, epigenetic mechanisms such as DNA methylation and microRNAs have also been shown to regulate BDNF expression [4,5].
At the protein level, BDNF is first synthesized as a precursor form pro-BDNF, which is then processed by proteases to generate mature BDNF. The two have distinct functional differences: pro-BDNF usually mediates synaptic weakening or apoptosis through the p75NTR receptor, while mature BDNF promotes neuronal survival and synaptic enhancement through the TrkB receptor [7]. Therefore, the dynamic balance between pro-BDNF and mature BDNF is considered a key mechanism regulating neuroplasticity.
In addition, the BDNF gene Val66Met (rs6265) polymorphism affects the intracellular transport and secretion efficiency of BDNF and is associated with the risk of various neurological diseases [6]. Structural biology studies further indicate that mutations in key residues of the BDNF protein may alter its conformational stability and receptor binding ability, thereby affecting signal transduction efficiency [8].
1.2 BDNF-TrkB Signaling Pathway
The main biological functions of BDNF are mediated by the high-affinity receptor TrkB (tropomyosin receptor kinase B). Binding of BDNF to TrkB induces receptor dimerization and autophosphorylation of tyrosine residues, thereby activating multiple downstream signaling pathways including PI3K/Akt, MAPK/ERK, and PLC-γ pathways [9,10]. These signaling networks collectively regulate neuronal survival, axon growth, and synaptic plasticity.
1.2.1 MAPK/ERK Pathway
The MAPK/ERK pathway is one of the most classical downstream pathways of BDNF-TrkB signaling. After phosphorylation, the TrkB receptor activates the Ras-Raf-MEK-ERK cascade through adaptor proteins such as Shc and Grb2. Activated ERK enters the nucleus and phosphorylates various transcription factors, such as CREB, thereby regulating neuronal differentiation and synapse-related gene expression.
This pathway plays important roles in dendrite growth, synapse formation, and long-term memory formation, and is the key molecular basis for BDNF regulation of learning and memory.
1.2.2 PI3K-Akt Pathway
The PI3K-Akt signaling pathway is mainly involved in cell survival and anti-apoptotic regulation. TrkB activation recruits PI3K and promotes Akt phosphorylation. Akt further phosphorylates proteins such as Bad and GSK-3β, thereby inhibiting apoptosis and enhancing neuronal survival [10].
In various neurodegenerative disease models, activation of the PI3K-Akt pathway is usually associated with neuroprotective effects, and is therefore considered one of the important mechanisms of BDNF-mediated neuroprotection.
1.2.3 PLC-γ Pathway
The PLC-γ pathway is mainly involved in intracellular calcium signal regulation. TrkB activation phosphorylates PLC-γ, which catalyzes the hydrolysis of PIP2 to produce IP3 and DAG. IP3 promotes Ca²⁺ release from the endoplasmic reticulum, while DAG activates protein kinase C (PKC). These signals collectively regulate neurotransmitter release and synaptic plasticity.
PLC-γ signaling plays an important role in synaptic plasticity processes such as long-term potentiation (LTP).
1.2.4 p75NTR Signaling Pathway
In addition to TrkB, the BDNF precursor pro-BDNF also functions through the low-affinity receptor p75NTR. p75NTR is usually associated with neuronal apoptosis, axon pruning, and synaptic weakening. This receptor regulates cell fate through signaling pathways such as JNK and NF-κB.
Therefore, the balance between the pro-BDNF/p75NTR and mBDNF/TrkB signaling axes is considered a key factor determining neuronal survival and synaptic plasticity.
2. BDNF-Related Diseases
2.1 Neurodegenerative Diseases
In neurodegenerative diseases such as Alzheimer's disease and Parkinson's disease, BDNF expression is generally decreased, which is closely related to neuronal loss and synaptic dysfunction. Reduced BDNF weakens the TrkB signaling pathway, thereby reducing neuronal survival signals and accelerating the neurodegenerative process.
In Alzheimer's disease models, decreased BDNF expression is closely associated with impaired hippocampal synaptic plasticity and cognitive decline. Restoring BDNF signaling can improve synaptic function and reduce cognitive impairment to a certain extent.
2.2 Psychiatric Disorders
BDNF also plays an important role in psychiatric disorders such as depression, anxiety, and schizophrenia. Studies have shown that serum BDNF levels are usually decreased in patients with depression, and antidepressant treatment can partially restore its expression.
BDNF participates in mood regulation by modulating hippocampal neurogenesis and synaptic plasticity, so abnormal expression may lead to dysfunction of mood regulation.
2.3 Nerve Injury
In conditions such as stroke, spinal cord injury, and traumatic brain injury, BDNF expression changes dynamically. BDNF participates in neurological function recovery by promoting axon regeneration, synapse reconstruction, and neural network remodeling.
Experimental studies have shown that increasing BDNF levels can promote nerve regeneration and improve neurological function recovery.
2.4 Pain and Drug Dependence
BDNF plays an important role in chronic pain and the formation of opioid tolerance. At the spinal cord level, BDNF enhances synaptic transmission and regulates pain signal transduction.
In morphine tolerance models, spinal BDNF expression is significantly increased, and intervention in BDNF signaling can partially alleviate the tolerance phenomenon [1]. These studies suggest that BDNF may be an important molecular target for pain regulation and drug dependence research.
2.5 Neurodevelopmental Disorders
BDNF participates in neuronal migration, axon guidance, and synapse formation during neurodevelopment. Therefore, abnormal expression may affect neural network formation and lead to neurodevelopmental disorders.
Recent studies have found that BDNF is associated with autism spectrum disorders and attention deficit hyperactivity disorder, particularly abnormal conversion of pro-BDNF to mature BDNF may affect synaptic plasticity and participate in developmental abnormalities [7].
3. Latest Research Progress of BDNF-Targeted Drugs
BDNF is a key molecule promoting neuronal survival and plasticity, and its targeted drug development has covered a variety of neuropsychiatric diseases. Currently, laquinimod has been approved for multiple sclerosis, and multiple clinical studies are exploring the efficacy of BDNF modulators in mental disorders such as depression and bipolar disorder. Preclinical projects are widely deployed for Alzheimer's disease, Parkinson's disease, glaucoma, and central nervous system injury, involving multiple technical routes such as small molecule chemicals, gene therapy, and traditional Chinese medicine, showing a diversified development trend overall. Some of the research pipelines are summarized in the table below:
| Drug Name
| Type |
Indications |
Development Stage |
Research Institution |
| Laquinimod |
Small molecule modulator |
Multiple sclerosis |
Approved |
Teva Pharmaceuticals |
| TrkB agonist |
Small molecule compound |
Depression/Alzheimer's disease |
Phase 2 |
Multiple pharmaceutical companies |
| BDNF gene therapy |
Gene therapy |
Parkinson's disease/Glaucoma |
Phase 1 |
Biotechnology companies |
| 7,8-Dihydroxyflavone |
Small molecule TrkB agonist |
Alzheimer's disease |
Preclinical |
Multiple research institutions |
| Active ingredients of traditional Chinese medicine (baicalin, ginsenoside Rb1, etc.) |
Natural products |
Depression/Nerve injury |
Preclinical |
Domestic research institutes |
| BDNF nano-delivery system |
Nano preparation |
Central nervous system injury |
Preclinical |
University collaboration projects |
(Data as of March 11, 2026, from synapse)
4. BDNF Research Tools Recommendation: Selection Guide for Recombinant Proteins, Antibodies, and ELISA Kits
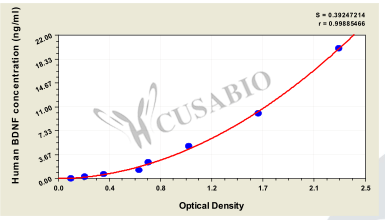
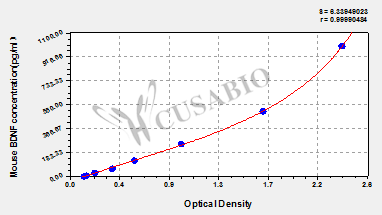
As the most widely distributed and deeply studied neurotrophic factor in the central nervous system, BDNF is a core molecule linking neuroplasticity, cognitive function, and the pathological mechanisms of various neuropsychiatric diseases. Regulation of its signaling pathway has become a strategic target for the treatment of refractory diseases such as Alzheimer's disease and depression. CUSABIO provides BDNF recombinant proteins, antibodies, and ELISA kits to support your mechanistic research and targeted drug development.
● BDNF Recombinant Proteins
● BDNF Antibodies
● BDNF ELISA Kits
References
[1] Zahra Khoshdel, Somayeh Ahmadpour Jirandeh, Mohammad Ali Takhshid, Farideh Jalali Mashayekhi, Shahla Shojaei, Ali Akbar Owji.(2019). The BDNF Protein and its Cognate mRNAs in the Rat Spinal Cord during Amylin-induced Reversal of Morphine Tolerance.
[2] Eli-Eelika Esvald, Jürgen Tuvikene, Andra Moistus, Käthy Rannaste, Susann Kõomägi, Tõnis Timmusk.(2022). Differential Regulation of the BDNF Gene in Cortical and Hippocampal Neurons.
[3] R Chow, J M Wessels, W G Foster.(2020). Brain-derived neurotrophic factor (BDNF) expression and function in the mammalian reproductive Tract.
[4] Te Zhao, Lian-Hua Piao, Dan-Ping Li, Shi-Han Xu, Shu-Yi Wang, Hai-Bo Yuan, Chun-Xiao Zhang.(2023). BDNF gene hydroxymethylation in hippocampus related to neuroinflammation-induced depression-like behaviors in mice.
[5] Gilmara Gomes De Assis, Eugenia Murawska-Ciałowicz.(2024). BDNF Modulation by microRNAs: An Update on the.
[6] Iwona Przybylska, Jarosław Marusiak, Beata Toczyłowska, Adam Stępień, Bogdan Brodacki, Józef Langfort, Małgorzata Chalimoniuk.(2024). Association between the Val66Met (rs6265) polymorphism of the brain-derived neurotrophic factor (BDNF) gene, BDNF protein level in the blood and the risk of developing early‑onset Parkinson's disease.
[7] Feng Yang, He You, Toshiyuki Mizui, Yasuyuki Ishikawa, Keizo Takao, Tsuyoshi Miyakawa, Xiaofei Li, Ting Bai, Kun Xia, Lingling Zhang, Dizhou Pang, Yiran Xu, Changlian Zhu, Masami Kojima, Bai Lu.(2024). Inhibiting proBDNF to mature BDNF conversion leads to ASD-like phenotypes in vivo.
[8] V M Datta Darshan, Natarajan Arumugam, Abdulrahman I Almansour, Venketesh Sivaramakrishnan, Subbarao Kanchi.(2024). In silico energetic and molecular dynamic simulations studies demonstrate potential effect of the point mutations with implications for protein engineering in BDNF.
[9] Peiyuan Huang, Aofei Liu, Yutong Song, Jen M Hope, Bianxiao Cui, Liting Duan.(2020). Optical Activation of TrkB Signaling.
[10] Si-Rui Sun, Jia-Ning Zhao, Peng-Wei Bi, Hui-Ying Zhang, Guang-Xiang Li, Jiao-Zhao Yan, Yun-Feng Li, Yong-Yu Yin, Hao Cheng.(2025). Pharmacologically activating BDNF/TrkB signaling exerted rapid-acting antidepressant-like effects through improving synaptic plasticity and neuroinflammation.
CUSABIO team. BDNF/Brain-Derived Neurotrophic Factor: From Neuroplasticity Regulation to Core Target for Neuropsychiatric Disease Therapy. https://www.cusabio.com/c-21299.html

-SDS.jpg)



Comments
Leave a Comment