[1] Anna Zhigun, Mabel Lizzy Rajendran.(2023). Modelling non-local cell-cell adhesion: a multiscale approach.
[2] XinXin Du, Ido Lavi, Michael J. Shelley.(2025). Collective multicellular patterns arising from cadherin-linked cytoskeletal domains.
[3] Virendra Singh, A. Singh, I. Sharma, L. Singh, J. Sharma, B. B. Borthakar, A. Rai, A. Kataki, S. Kapur, S. Saxena.(2019). Epigenetic deregulations of Wnt/β-catenin and transforming growth factor beta-Smad pathways in esophageal cancer: Outcome of DNA methylation.
[4] Preeti Sahu, Daniel M. Sussman, Matthias Rubsam, Aaron F. Mertz, Valerie Horsley, Eric R. Dufresne, Carien M. Niessen, M. Cristina Marchetti, M. Lisa Manning, J. M. Schwarz.(2019). Small-scale demixing in confluent biological tissues.
[5] C. Hakkaart, L. Ellison-Loschmann, R. Day, A. Sporle, Jonathan Koea, Pauline Harawira, Soo Cheng, M. Gray, Tracey Whaanga, N. Pearce, P. Guilford.(2018). Germline CDH1 mutations are a significant contributor to the high frequency of early-onset diffuse gastric cancer cases in New Zealand Māori.
[6] I. Chen, Lesley Mathews-Greiner, Dandan Li, A. Abisoye-Ogunniyan, Satyajit Ray, Yansong Bian, V. Shukla, Xiaohu Zhang, Rajarashi Guha, Craig J. Thomas, B. Gryder, A. Zacharia, J. Beane, S. Ravichandran, M. Ferrer, U. Rudloff.(2017). Transcriptomic profiling and quantitative high-throughput (qHTS) drug screening of CDH1 deficient hereditary diffuse gastric cancer (HDGC) cells identify treatment leads for familial gastric cancer.
[7] Nicola Bougen-Zhukov, Lyvianne Decourtye-Espiard, Wilson Mitchell, Kieran Redpath, J. Perkinson, T. Godwin, M. Black, P. Guilford.(2022). E-Cadherin-Deficient Cells Are Sensitive to the Multikinase Inhibitor Dasatinib.
[8] T. Godwin, S. Kelly, Tom Brew, Nicola Bougen-Zhukov, A. Single, Augustine Chen, Cassie E Stylianou, L. Harris, Sophie K Currie, Bryony J. Telford, Henry Beetham, G. Evans, M. Black, P. Guilford.(2018). E-cadherin-deficient cells have synthetic lethal vulnerabilities in plasma membrane organisation, dynamics and function.
[9] G. Corso, M. Intra, C. Trentin, P. Veronesi, V. Galimberti.(2016). CDH1 germline mutations and hereditary lobular breast cancer.
[10] C. São José, J. Garcia-Pelaez, Marta Ferreira, O. Arrieta, Ana André, Nelson Martins, Samantha Solís, B. Martínez-Benítez, M. Ordoñez-Sánchez, M. Rodríguez-Torres, Anna K. Sommer, Iris B A W Te Paske, C. Caldas, M. Tischkowitz, María Teresa Tusié, Stefan Gabriel Sérgio Richarda M. Gareth Susana José Luzi Aretz Capella Castedo de Voer Evans Fernandes Garc, S. Aretz, G. Capellá, S. Castedo, Richarda M. de Voer, Gareth Evans, Susana Fernandes, J. Garcia-Pelaez, L. Garrido, E. Holinski-Feder, N. Hoogerbrugge, D. Huntsman, A. Jahn, C. Kets, A. Laner, M. Ligtenberg, Andrea Meinhardt, A. Mensenkamp, Carla Oliveira, S. Peters, I. Quintana, E. Schröck, Anna K. Sommer, I. Spier, L. Spruijt, V. Steinke-Lange, I. T. Paske, M. Tischkowitz, L. Valle, R. S. van der Post, Y. V. van Herwaarden, W. V. van Zelst-Stams, D. William, N. Hoogerbrugge, German Demidov, Richarda M. de Voer, S. Laurie, Carla Oliveira.(2023). Combined loss of CDH1 and downstream regulatory sequences drive early-onset diffuse gastric cancer and increase penetrance of hereditary diffuse gastric cancer.
[11] G. Tedaldi, C. Molinari, C. São José, Rita Barbosa-Matos, A. André, R. Danesi, V. Arcangeli, M. Ravegnani, L. Saragoni, P. Morgagni, F. Rebuzzi, M. Canale, S. Pignatta, Elisa Ferracci, G. Martinelli, G. Ranzani, Carla Oliveira, D. Calistri, P. Ulivi.(2021). Genetic and Epigenetic Alterations of CDH1 Regulatory Regions in Hereditary and Sporadic Gastric Cancer.
[12] Yin Tong, P. Cheng, Chung Sze Or, Sarah S K Yue, H. Siu, S. Ho, S. Law, W. Tsui, D. Chan, Stephanie Ma, Siu Po Lee, Annie S. Y. Chan, April S Chan, Shui Wa Yun, Ho Sang Hui, S. Yuen, S. Leung, Helen H. N. Yan.(2022). Escape from cell-cell and cell-matrix adhesion dependence underscores disease progression in gastric cancer organoid models.
[13] D. Xie, Yiyu Chen, Xue Wan, Jingyuan Li, Qin Pei, Yanan Luo, Jinbo Liu, Ting Ye.(2022). The Potential Role of CDH1 as an Oncogene Combined With Related miRNAs and Their Diagnostic Value in Breast Cancer.
[14] Jiaming Gao, Yunying Yao, Zaiqi Wang, Baoyuan Zhang, R. Ren.(2023). Abstract 5821: Synergism of FAK and ROS1 inhibitors in the treatment of CDH1-deficient cancers mediated by FAK-YAP-TRX signaling.
[15] Da-Tian Bau, Ting-Yuan Liu, Jai-Sing Yang, William Tzu-Liang Chen, Chia‐Wen Tsai, Wen‐Shin Chang, Tao-Wei Ke, Chi-Chou Liao, Yu-Chia Chen, Yen-Ting Chang, Fuu-Jen Tsai.(2024). Characterizing Genetic Susceptibility to Colorectal Cancer in Taiwan Through Genome‐Wide Association Study.
[16] Di Wang, Biao Xie.(2022). Prognostic and tumor immunity implication of inflammatory bowel disease-associated genes in colorectal cancer.
[17] H. Ishiguro, T. Wakasugi, Yukio Terashita, N. Sakamoto, Tatsuya Tanaka, K. Mizoguchi, H. Sagawa, T. Okubo, H. Takeyama.(2016). Decreased expression of CDH1 or CTNNB1 affects poor prognosis of patients with esophageal cancer.
[18] L. Hämäläinen, Y. Soini, S. Pasonen-Seppänen, M. Siponen.(2019). Alterations in the expression of EMT-related proteins claudin-1, -4 and -7, E-cadherin, TWIST1 and ZEB1 in oral lichen planus..
[19] J. Figueiredo, F. Mercadillo, Soraia Melo, A. Barroso, Margarida Gonçalves, J. Diaz-Tasende, Patrícia Carneiro, L. Robles, F. Colina, C. Ibarrola, J. Perea, Eurico Morais-de-Sá, R. Seruca, M. Urioste.(2021). Germline CDH1 G212E Missense Variant: Combining Clinical, In Vitro and In Vivo Strategies to Unravel Disease Burden.
[20] A. F. Ismail, Sevil Oskay Halacli, N. Babteen, M. De Piano, T. Martin, W. Jiang, Muhammad S. Khan, P. Dasgupta, C. Wells.(2017). PAK5 mediates cell: cell adhesion integrity via interaction with E-cadherin in bladder cancer cells..
[21] F. Benthani, F. Benthani, D. Herrmann, D. Herrmann, P. N. Tran, P. N. Tran, L. Pangon, L. Pangon, M. Lucas, A. Allam, N. Currey, S. Al‐Sohaily, Marc Giry-Laterrière, J. Warusavitarne, P. Timpson, P. Timpson, M. R. Kohonen-Corish, M. R. Kohonen-Corish, M. R. Kohonen-Corish.(2018). ‘MCC’ protein interacts with E-cadherin and β-catenin strengthening cell–cell adhesion of HCT116 colon cancer cells.
[22] Sònia Bassagañas, S. Carvalho, A. M. Dias, Marta Pérez-Garay, M. R. Ortiz, J. Figueras, C. Reis, S. Pinho, R. Peracaula.(2014). Pancreatic Cancer Cell Glycosylation Regulates Cell Adhesion and Invasion through the Modulation of α2β1 Integrin and E-Cadherin Function.
[23] M. Katayama, S. Hirai, M. Yasumoto, K. Nishikawa, S. Nagata, M. Otsuka, K. Kamihagi, I. Kato.(1994). Soluble fragments of e-cadherin cell-adhesion molecule increase in urinary-excretion of cancer-patients, potentially indicating its shedding from epithelial tumor-cells..
[24] Charles de Ponthaud, Solafah Abdalla, M. Belot, Xiaojian Shao, Christophe Penna, A. Brouquet, Pierre Bougnères.(2024). Increased CpG methylation at the CDH1 locus in inflamed ileal mucosa of patients with Crohn disease.
[25] Asia Asiaf, S. T. Ahmad, S. Aziz, A. Malik, Zubaida Rasool, A. Masood, M. Zargar.(2014). Loss of expression and aberrant methylation of the CDH1 (E-cadherin) gene in breast cancer patients from Kashmir..
[26] Hang Song, Yao-quan Zhang, Na Liu, Sheng Zhao, Y. Kong, Liudi Yuan.(2016). miR-92a-3p Exerts Various Effects in Glioma and Glioma Stem-Like Cells Specifically Targeting CDH1/β-Catenin and Notch-1/Akt Signaling Pathways.
[27] M. Bagherpour, Kamelia Gharibzad, H. Rassi.(2018). Association of CDH1 and TERT Single-Nucleotide Polymorphisms with Susceptibility to Familial Breast Cancer Risk..
[28] Geonhui Lee, Claudia Wong, Anna Cho, Junior J West, Ashleigh J. Crawford, Gabriella C. Russo, B. R. Si, Jungwoo Kim, Lauren Hoffner, Cholsoon Jang, Moonjung Jung, Robert D Leone, K. Konstantopoulos, Andrew J Ewald, Denis Wirtz, Sangmoo Jeong.(2024). E-cadherin Induces Serine Synthesis to Support Progression and Metastasis of Breast Cancer..
[29] I. O. Osman, Clémence Garrec, Gabriel Augusto Pires de Souza, A. Zarubica, Djamal Brahim Belhaouari, J. Baudoin, H. Lepidi, J. Mege, B. Malissen, B. Scola, C. Devaux.(2022). Control of CDH1/E-Cadherin Gene Expression and Release of a Soluble Form of E-Cadherin in SARS-CoV-2 Infected Caco-2 Intestinal Cells: Physiopathological Consequences for the Intestinal Forms of COVID-19.
[30] L. Thorstensen, G. E. Lind, T. Løvig, C. B. Diep, G. Meling, T. Rognum, R. Lothe.(2005). Genetic and epigenetic changes of components affecting the WNT pathway in colorectal carcinomas stratified by microsatellite instability..
[31] Hong Liu, Yan Ma, Hong-wei He, Wu-li Zhao, R. Shao.(2017). SPHK1 (sphingosine kinase 1) induces epithelial-mesenchymal transition by promoting the autophagy-linked lysosomal degradation of CDH1/E-cadherin in hepatoma cells.
[32] Xiao Tan, Wenbin Chen, D. Lv, Tao-wei Yang, Kai-hui Wu, Li-bin Zou, Junqi Luo, Xu-min Zhou, Guo-chang Liu, F. Shu, X. Mao.(2020). LncRNA SNHG1 and RNA binding protein hnRNPL form a complex and coregulate CDH1 to boost the growth and metastasis of prostate cancer.
[33] Yan He, Qimin Zhang, Yuan-Xiao Chen, Yingjian Wu, Yuan Quan, Weihua Chen, Jing Yao, Peijing Zhang.(2023). ZHX2 deficiency enriches hybrid MET cells through regulating E-cadherin expression.

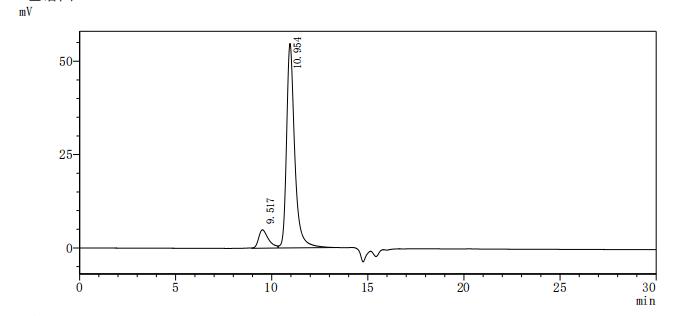
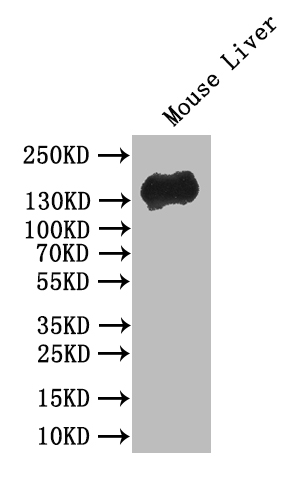
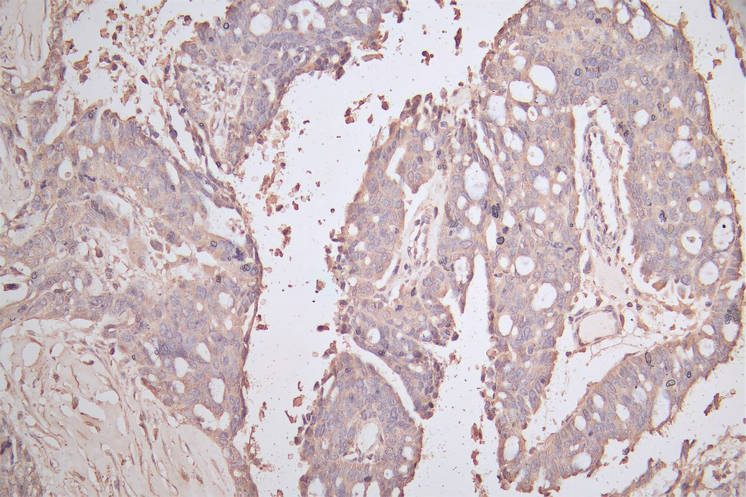
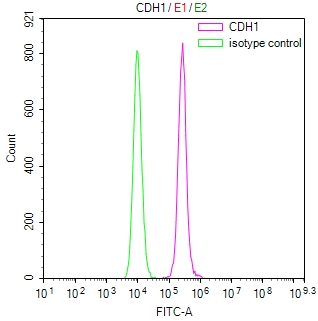


Comments
Leave a Comment