[1] Jonathan H Esensten, Ynes A Helou, Gaurav Chopra, Arthur Weiss, Jeffrey A Bluestone.(2016). CD28 Costimulation: From Mechanism to Therapy.
[2] Songna Wang, Pinliang Hu, Xuyao Zhang, Jiajun Fan, Jing Zou, Weidong Hong, Xuan Huang, Danjie Pan, Huaning Chen, Dianwen Ju, Yi Zhun Zhu, Li Ye.(2025). Recombinant CD80 fusion protein combined with discoidin domain receptor 1 inhibitor for cancer treatment.
[3] Masashi Watanabe, Sofia Celli, Farrah A Alkhaleel, Richard J Hodes.(2022). Antigen-presenting T cells provide critical B7 co-stimulation for thymic iNKT cell development via CD28-dependent trogocytosis.
[4] Wei Yang, Weiling Pan, Shuokai, Nicola Trendel, Shutan Jiang, Feng Xiao, Manman Xue, Wei Wu, Zeli Peng, Xiaoxi Li, Hongbin Ji, Xiaolong Liu, Hai Jiang, Haopeng Wang, Hongbin Shen, Omer Dushek, Hua Li, Chenqi Xu.(2017). Dynamic regulation of CD28 conformation and signaling by charged lipids and ions.
[5] Jeffrey Comer, Molly Bassette, Riley Burghart, Mayme Loyd, Susumu Ishiguro, Ettayapuram Ramaprasad Azhagiya Singam, Ariela Vergara-Jaque, Ayaka Nakashima, Kengo Suzuki, Brian V Geisbrecht, Masaaki Tamura.(2021). Beta-1,3 Oligoglucans Specifically Bind to Immune Receptor CD28 and May Enhance T Cell Activation.
[6] Kumarkrishna Raychaudhuri, Rohita Rangu, Alison Ma, Neriah Alvinez, Andy D Tran, Sandeep Pallikkuth, Katherine M McIntire, Joseph A Garvey, Jason Yi, Lawrence E Samelson.(2024). CD28 shapes T cell receptor signaling by regulating Lck dynamics and ZAP70 activation.
[7] Jennifer A. Rohrs, Elizabeth L. Siegler, Pin Wang, Stacey D. Finley.(2020). ERK Activation in CAR T Cells Is Amplified by CD28-Mediated Increase in CD3ζ Phosphorylation.
[8] Anuja Bhatta, Marcia A Chan, Stephen H Benedict.(2021). Transcription factor activation and protein phosphorylation patterns are distinct for CD28 and ICAM-1 co-stimulatory molecules.
[9] Xuelian Xiang, Nan Cao, Feiyue Chen, Long Qian, Yifei Wang, Yunmao Huang, Yunbo Tian, Danning Xu, Wanyan Li.(2020). Polysaccharide of Atractylodes macrocephala Koidz (PAMK) Alleviates Cyclophosphamide-induced Immunosuppression in Mice by Upregulating CD28/IP3R/PLCγ-1/AP-1/NFAT Signal Pathway.
[10] Brian Tomkowicz, Eileen S. Walsh, Adam Cotty, Raluca Verona, Nina Chi-Sabins, Fred M. Kaplan, Sandy Santulli-Marotto, Chen-Ni Chin, Jill M. Mooney, Russell B. Lingham, Michael Naso, Timothy J. McCabe.(2015). TIM-3 Suppresses Anti-CD3/CD28-Induced TCR Activation and IL-2 Expression through the NFAT Signaling Pathway.
[11] Sadiye Amcaoglu Rieder, Jingya Wang, Natalie White, Ariful Qadri, Catherine Menard, Geoffrey Stephens, Jodi L Karnell, Christopher E Rudd, Roland Kolbeck.(2021). B7-H7 (HHLA2) inhibits T-cell activation and proliferation in the presence of TCR and CD28 signaling.
[12] Enfu Hui, Jeanne Cheung, Jing Zhu, Xiaolei Su, Marcus J Taylor, Heidi A Wallweber, Dibyendu K Sasmal, Jun Huang, Jeong M Kim, Ira Mellman, Ronald D Vale.(2017). T cell costimulatory receptor CD28 is a primary target for PD-1-mediated inhibition.
[13] G Aaron Holling, Colin A Chavel, Anand P Sharda, Mackenzie M Lieberman, Caitlin M James, Shivana M Lightman, Jason H Tong, Guanxi Qiao, Tiffany R Emmons, Thejaswini Giridharan, Shengqi Hou, Andrew M Intlekofer, Richard M Higashi, Teresa W M Fan, Andrew N Lane, Kevin H Eng, Brahm H Segal, Elizabeth A Repasky, Kelvin P Lee, Scott H Olejniczak.(2024). CD8+ T cell metabolic flexibility elicited by CD28-ARS2 axis-driven alternative splicing of PKM supports antitumor immunity.
[14] Marianne Strazza, Inbar Azoulay-Alfaguter, Bryan Dun, Jairo Baquero-Buitrago, Adam Mor.(2015). CD28 inhibits T cell adhesion by recruiting CAPRI to the plasma membrane.
[15] Masashi Watanabe, Ying Lü, Michael Breen, Richard J. Hodes.(2020). B7-CD28 co-stimulation modulates central tolerance via thymic clonal deletion and Treg generation through distinct mechanisms.
[16] Yinghao Zhao, Wei Yang, Yuanyuan Huang, Ranji Cui, Xiangyan Li, Bingjin Li.(2018). Evolving Roles for Targeting CTLA-4 in Cancer Immunotherapy.
[17] Shunsuke Fukuyo, Shingo Nakayamada, Shigeru Iwata, Satoshi Kubo, Kazuyoshi Saito, Yoshiya Tanaka.(2017). Abatacept therapy reduces CD28+CXCR5+ follicular helper-like T cells in patients with rheumatoid arthritis.
[18] Alice O Kamphorst, Andreas Wieland, Tahseen Nasti, Shu Yang, Ruan Zhang, Daniel L Barber, Bogumila T Konieczny, Candace Z Daugherty, Lydia Koenig, Ke Yu, Gabriel L Sica, Arlene H Sharpe, Gordon J Freeman, Bruce R Blazar, Laurence A Turka, Taofeek K Owonikoko, Rathi N Pillai, Suresh S Ramalingam, Koichi Araki, Rafi Ahmed.(2017). Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent.
[19] Wei Huff, Marpe Bam, Jack Shireman, Jae Hyun Kwon, Leo Song, Sharlé Newman, Aaron Cohen‐Gadol, Scott Shapiro, Tamara Jones, Kelsey Fulton, Sheng Liu, Hiromi Tanaka, Yunlong Liu, Jun Wan, Mahua Dey.(2021). Aging- and Tumor-Mediated Increase in CD8+CD28− T Cells Might Impose a Strong Barrier to Success of Immunotherapy in Glioblastoma.
[20] Zhen Yang, Xinpeng Liu, Jun Zhu, Yangyang Chai, Boyi Cong, Bo Li, Wanfeng Gao, Ye Hu, Mingyue Wen, Yanfang Liu, Li Fu, Xuetao Cao.(2025). Inhibiting intracellular CD28 in cancer cells enhances antitumor immunity and overcomes anti-PD-1 resistance via targeting PD-L1.
[21] Ryan C Augustin, Riyue Bao, Jason J Luke.(2023). Targeting Cbl-b in cancer immunotherapy.
[22] Xuexiao Jin, Qin Xu, Chengfei Pu, Kaixiang Zhu, Cheng Lu, Yu Jiang, Lei Xiao, Yongmei Han, Linrong Lu.(2021). Therapeutic efficacy of anti-CD19 CAR-T cells in a mouse model of systemic lupus erythematosus.
[23] Ling Qin, Jing Xie, Zhifeng Qiu, Wei Cao, Yang Jiao, Jean‐Pierre Routy, Taisheng Li.(2016). Aging of immune system: immune signature from peripheral blood lymphocyte subsets in 1068 healthy adults.


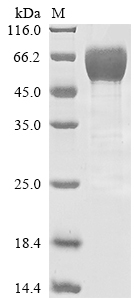
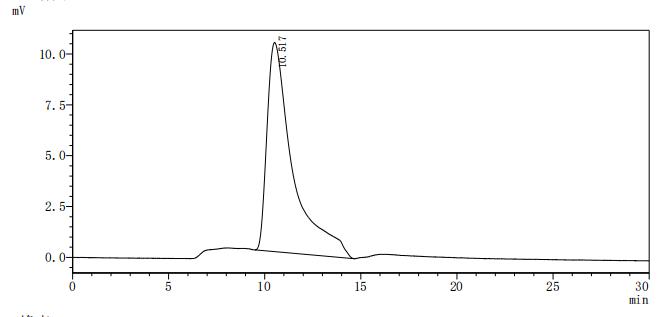
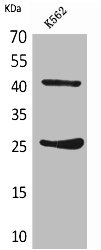
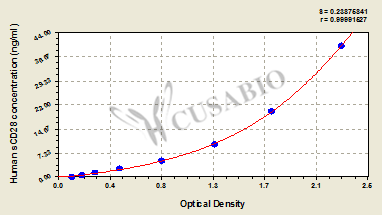


Comments
Leave a Comment