CCN2 (also known as connective tissue growth factor, CTGF) is an important multifunctional matricellular protein that plays a key role in tissue development, homeostasis maintenance, fibrosis, cancer and various pathological remodeling processes. Existing studies have shown that CCN2 is not a simple "growth factor" in the traditional sense, but an important signaling node that regulates cell proliferation, differentiation, migration, adhesion and tissue remodeling through interactions with the extracellular matrix, receptors and various ligands [1,2]. Its activity has obvious structure-dependent and microenvironment-dependent characteristics: full-length CCN2 may exist as an inactive precursor, while the C-terminal fragment produced by proteolysis and its homodimer are more important active forms [5]. CCN2 is closely related to multiple signaling pathways such as TGF-β/Smad, FAK, EGFR, MAPK/ERK, PI3K/AKT, and Hippo/YAP, and exhibits complex roles in fibrosis, cancer, vascular diseases, metabolic abnormalities, inflammation and developmental disorders [3-5]. Overall, CCN2 is an important molecule in disease mechanism research and a potential translational research target, but its different active forms, tissue environment dependence and the balance relationship among family members still need further study.
1. Research Background and Biological Functions of CCN2 (CTGF)
The CCN protein family is a class of multifunctional matricellular proteins secreted into the extracellular matrix microenvironment, which are mainly involved in organ development, tissue homeostasis maintenance and various disease processes by regulating the interactions between cells and between cells and the extracellular matrix (ECM) [1]. In this family, CCN2, also known as CTGF, is currently the most extensively studied member.
Although the name "connective tissue growth factor" easily leads to the understanding of CCN2 as a traditional growth factor, increasing studies suggest that CCN2 is more appropriately defined as a multifunctional matricellular protein. Its role is not limited to promoting growth, but is widely involved in multiple processes such as cell proliferation, differentiation, migration, adhesion, ECM synthesis and tissue remodeling [2-4]. Meanwhile, there are complex regulatory relationships among CCN family members. For example, CCN2 and CCN3 can exhibit a "Yin-Yang" balance in some tissues, suggesting that their relative expression status may affect tissue homeostasis and disease progression [1].
From the perspective of disease research, the most concerned fields of CCN2 mainly include fibrosis and cancer. Existing studies have shown that CCN2 is often abnormally expressed in various fibrotic diseases and malignant tumors, and is associated with enhanced tissue remodeling, active stromal reaction or disease progression [2,5]. For example, in pancreatic ductal adenocarcinoma, CCN2 can promote stromal cell infiltration and enhance the interaction between tumor and stroma, and its high expression is usually associated with poor prognosis [2]. Therefore, CCN2 is not only an important node in basic research, but also has gradually become a hotspot molecule in disease mechanism and potential intervention directions.
2. Structural Characteristics and Basic Biological Functions of CCN2
CCN2 is a cysteine-rich secreted protein, and its molecular structure has obvious modular characteristics. It consists of four conserved domains, namely the IGFBP domain, VWC domain, TSP1 repeat sequence domain and CT domain. This structure enables CCN2 to interact with a variety of molecules, and also provides a basis for its extensive and complex biological functions.
Existing studies suggest that the activity of CCN2 cannot be simply equated with the full-length protein itself. Full-length CCN2 may not be in a fully activated state, but more like a precursor molecule with certain self-inhibitory characteristics; after proteolytic cleavage, part of its activity is truly released [5]. Among them, the C-terminal fragment composed of domain III and domain IV is considered to carry the important biological activity of CCN2 [5]. Further studies have also found that these C-terminal fragments can form homodimers, which are significantly stronger than monomers in activating intracellular kinase cascade reactions, and more effectively promote fibroblast migration, focal adhesion assembly, osteoclast differentiation and breast cancer spheroid formation [5]. This indicates that CCN2 is not a simple "monomorphic protein", but a complex signaling molecule jointly regulated by processing status, conformational changes and domain combinations.
In some disease contexts, different modules of CCN2 may also have relatively independent functions. For example, module 4 encoded by exon 5 of Ccn2 has a specific role in renal fibrosis. Relevant studies have shown that after knocking out this module, the degree of interstitial fibrosis in various renal fibrosis models is significantly reduced [6]. This result indicates that the understanding of CCN2 cannot stay at the overall expression level, and further attention needs to be paid to its specific domains and active fragments.
Under physiological conditions, CCN2 is involved in processes such as development, tissue repair, wound healing, angiogenesis and cell adhesion [2]. It regulates cell behavior through interactions with ECM, receptors and other molecular partners, so its function has obvious tissue dependence and microenvironment dependence. For this reason, CCN2 may exhibit biological effects of different degrees or even different directions in different organs and different disease stages.
3. Mechanism of Action of CCN2: How Does It Regulate Cells and Tissue Microenvironment?
The core of the mechanism of action of CCN2 in cells and tissues is that it can affect both cell behavior and matrix environment simultaneously. As an important matricellular protein, CCN2 is not only involved in cell proliferation, migration and differentiation, but also directly affects the synthesis, deposition and remodeling of ECM. Therefore, it often appears in processes such as tissue repair, fibrosis, tumor stromal reaction and chronic pathological remodeling.
In fibrosis-related mechanisms, the most prominent role of CCN2 is to promote fibroblast activation, myofibroblast formation and ECM accumulation. Studies have shown that TGF-β can induce increased expression of CCN1 and CCN2 in dermal and gingival fibroblasts and promote the formation of fibrotic phenotypes. Meanwhile, ROS and NADPH oxidase are also involved in this process; the antioxidant N-acetylcysteine (NAC) and NOX inhibitors can inhibit TGF-β1-induced expression of CCN2 and α-SMA, indicating that CCN2 is located at the important node where TGF-β intersects with oxidative stress [7]. In other words, CCN2 is not just an accompanying molecule in the fibrosis process, but a key mediator that promotes the continuous amplification of fibrotic signals.
In addition to endogenous signals, environmental factors also affect CCN2-related responses. For example, vitamin C can enhance the expression of COL1A1, ACTA2 and COL4A1 under TGF-β1 stimulation, and promote the formation of α-SMA stress fibers, thereby further amplifying the pro-fibrotic effect [8]. This indicates that the CCN2-related mechanism is not an isolated pathway, but is regulated by metabolic status, redox environment and extracellular stimulation.
In addition, CCN2 is also directly involved in extracellular matrix remodeling and cell adhesion regulation. Its active fragment can enhance the assembly of focal adhesion complexes, affect the stability of the connection between cells and ECM, and further regulate cell migration, morphological maintenance and tissue remodeling [5]. Therefore, CCN2 is not only a bridge connecting intracellular signals and the external matrix environment, but also an important molecule that promotes tissue transformation from "normal repair" to "abnormal remodeling".
4. Key Signaling Pathways Regulated by CCN2
A key reason why CCN2 plays an important role in fibrosis, cancer and other diseases is that it is not a local molecule in a single pathway, but is deeply embedded in multiple important signaling networks, and plays the role of connection, amplification and integration among these networks.
4.1 TGF-β/Smad Pathway
The TGF-β/Smad pathway is currently the most clearly and extensively studied pathway related to CCN2. A large number of studies have shown that TGF-β can significantly induce CCN2 expression, and CCN2 will further enhance fibroblast activation, ECM deposition and myofibroblast formation, forming a pro-fibrotic positive feedback loop. That is to say, CCN2 is not a simple downstream response molecule, but will further amplify upstream stimulation, making tissue remodeling and fibrosis reactions more sustained and obvious [9].
This signaling axis is of representative significance in various fibrotic diseases. Whether in the skin, kidney, lung or other tissues, the TGF-β/Smad-CCN2 network is generally regarded as the core component of pro-fibrosis. Therefore, from mechanism research to target analysis, this pathway has always been the focus of understanding CCN2 function.
4.2 Hippo/YAP Pathway
The Hippo/YAP pathway is mainly related to mechanical stimulation, tissue tension, cell density and organ size regulation. The connection between CCN2 and this pathway indicates that it not only responds to chemical signals, but also responds to mechanical signals. Studies suggest that in the context of enhanced mechanotransduction or altered tissue tension, YAP activation can promote CCN2 expression; after CCN2 increases, it can further affect cell adhesion, migration and fibrotic reactions [10].
This is particularly important for chronic injury, scar formation and tumor stromal remodeling. Because these pathological processes are usually accompanied not only by changes in cytokines, but also by increased tissue stiffness, ECM remodeling and changes in the mechanical environment. CCN2 acts more like a "mechanical signal converter" here, converting tension changes in the tissue microenvironment into specific biological responses.
4.3 FAK Pathway
The FAK signaling pathway is closely related to cell adhesion, migration and cytoskeletal reorganization. As a key molecule in focal adhesion signaling, FAK can sense changes in the connection between cells and ECM, and regulate the mechanical adaptation and motor behavior of cells. Studies suggest that CCN2 can promote the assembly of focal adhesion complexes and enhance related kinase cascade reactions, thereby affecting cytoskeletal organization, migration ability and adhesion stability [6].
It is particularly noteworthy that the C-terminal active fragment of CCN2 and its dimer form are more obvious in promoting fibroblast migration and adhesion structure assembly [5]. This mechanism not only helps explain the role of CCN2 in wound repair and fibrosis, but also explains its function in tumor invasion and tissue remodeling.
4.4 EGFR, MAPK/ERK and PI3K/AKT Pathways
In cancer-related research, the interaction between CCN2 and pathways such as EGFR, MAPK/ERK and PI3K/AKT is also very important. EGFR is a classic pro-proliferation and pro-survival signaling axis, and the crosstalk between CCN2 and EGFR signaling provides a mechanistic basis for its involvement in tumor growth and invasion. By interacting with these pathways, CCN2 can enhance the proliferation ability, survival ability and adaptability to the surrounding matrix environment of tumor cells [11].
Meanwhile, MAPK/ERK and PI3K/AKT are also core pathways widely existing in various physiological and pathological processes. By affecting these downstream networks, CCN2 can further support cell migration, differentiation, proliferation and damage response. In the context of fibrosis, these pathways help maintain the continuous activation of fibroblasts; in the context of tumors, they help improve the survival and expansion ability of tumor cells.
4.5 Overall Characteristics of CCN2 Signal Regulation
Overall, CCN2 signal regulation has three characteristics. First, it can integrate multiple types of upstream stimuli, including growth factors, oxidative stress and mechanical stimuli. Second, it can amplify multiple types of downstream effects, including migration, adhesion, proliferation and matrix remodeling. Third, it is at the intersection of multiple pathways and has obvious network regulatory properties. Therefore, the role of CCN2 is not a linear one-way regulation, but more like a multi-level, multi-directional signal integration hub.
5. Research Progress of CCN2-Related Diseases
CCN2 is closely related to multiple types of diseases, especially in fibrotic diseases and cancer. At the same time, it is also involved in vascular diseases, bone-related lesions, metabolic abnormalities, inflammatory diseases, reproductive system diseases, developmental disorders and drug-related injuries. Overall, CCN2 is not a molecule unique to a certain disease, but a common node in various tissue remodeling processes.
5.1 CCN2 and Fibrotic Diseases
Fibrosis is one of the core directions in CCN2 research. Existing studies have shown that CCN2 is closely related to various diseases such as systemic sclerosis, pulmonary fibrosis, renal fibrosis, skin fibrosis, epidural fibrosis, intestinal fibrosis and cardiac fibrosis. In these diseases, CCN2 is usually involved in fibroblast activation, myofibroblast formation, increased ECM deposition and tissue stiffening, so it is widely regarded as a key molecule in the pro-fibrotic network [12].
Its importance is not only reflected in increased expression, but also in its substantive role in promoting the pathological process. CCN2 can work together with TGF-β, oxidative stress and mechanical stimulation to continuously prolong and amplify the local repair response, and finally turn from normal repair to pathological scarring. Especially in renal fibrosis, module 4 encoded by exon 5 of Ccn2 has been proven to have a specific pathogenic role; after knocking out this module, the degree of interstitial fibrosis in various renal fibrosis models is significantly reduced [6]. This result indicates that CCN2 is not only a marker molecule of fibrosis, but also a structural and functional unit with clear pathogenic significance.
5.2 CCN2 and Cancer
Compared with fibrosis, the role of CCN2 in cancer is more complex and more dependent on the specific context. Studies have shown that CCN2 can promote tumor progression in bladder cancer, liver cancer, pancreatic cancer, prostate cancer and some lymphomas [2,5,13]. Its tumor-promoting effect is not only directly acting on tumor cells themselves, but also often achieved by enhancing the interaction between tumor cells and the stroma, promoting migration and invasion, and shaping a supportive microenvironment.
For example, in pancreatic ductal adenocarcinoma, CCN2 can promote stromal cell infiltration and strengthen tumor-stroma interaction, which is closely related to poor prognosis [2]. This indicates that in some tumors, CCN2 acts more like a "tumor microenvironment amplifier", creating a more favorable environment for tumor progression by regulating stromal reaction and ECM status.
However, CCN2 cannot be simply classified as a "tumor-promoting molecule". In some tumor types or specific contexts, it may also exhibit the effect of inhibiting metastasis or limiting lesion expansion. For example, in some contexts of lung cancer, CCN2 has been observed to have the effect of inhibiting metastasis [14]. This suggests that the role of CCN2 in cancer is highly dependent on tumor type, cell origin, disease stage and microenvironment status. Therefore, in cancer research, CCN2 is more appropriately understood as a context-dependent tumor microenvironment regulator.
5.3 CCN2 and Vascular, Bone and Other Diseases
In addition to fibrosis and cancer, CCN2 is also related to diseases of the vascular system and skeletal system. In the vascular system, CCN2 is involved in the maintenance of smooth muscle cell phenotype and vascular wall homeostasis, and its abnormality can affect the progression of diseases such as aortic lesions [15]. In bone and connective tissue research, CCN2 is closely related to cell differentiation, tissue reconstruction and repair processes. Because it can regulate cell adhesion, matrix remodeling and mechanical signal integration, its role in these tissues is also of great significance [16].
In addition, CCN2 has also been studied in metabolic diseases, inflammatory diseases, reproductive system diseases, developmental disorders and drug-related cardiotoxicity [17]. These results further indicate that CCN2 is not a local molecule limited to a certain organ or disease type, but an important molecule involved in tissue remodeling and pathological adaptation across organs and disease types.
6. Research Progress of CCN2-Targeted Drug Development
Currently, the research and development of CCN2-targeted drugs has formed a complete pattern from preclinical to phase 3 clinical trials. The drug types cover monoclonal antibodies, siRNA and bispecific antibodies, etc., mainly focusing on the treatment of fibrotic diseases and cancer. With the participation of multiple institutions, its clinical translation prospect is receiving widespread attention.
|
Medications
|
Target
|
Types of medications
|
Indications under investigation
|
Research institutions
|
Highest R&D stage
|
|
Panreelumab
|
CTGF
|
Monoclonal antibodies
|
Locally Advanced Pancreatic Adenocarcinoma | Unresectable pancreatic cancer
|
Kyntra Bio, Inc. | Boehringer Ingelheim Pharma GmbH & Co., KG
|
Phase 3 clinical trial
|
|
SHR-1906
|
CTGF
|
Monoclonal antibodies
|
Idiopathic pulmonary fibrosis
|
Guangdong Hengrui Pharmaceutical Co., Ltd
|
Clinical phase 2
|
|
LEM-S401
|
CTGF
|
siRNA
|
Skin hypertrophic scarring
|
Lemonex Co., Ltd.
|
Clinical phase 1
|
|
OLX-701
|
CTGF
|
siRNA
|
Cirrhosis
|
OliX Pharmaceuticals, Inc.
|
Preclinical
|
|
IB-DMD(INNOBIOSCIENCE)
|
CTGF x NF-κB
|
Stem cell therapy
|
Duchenne muscular dystrophy
|
INNOBIOSCIENCE LLC
|
Preclinical
|
|
GenSciP111
|
CTGF
|
siRNA
|
Idiopathic pulmonary fibrosis
|
Changchun Kinsey Pharmaceutical Co., Ltd
|
Preclinical
|
|
OLX-201A
|
CTGF
|
siRNA
|
Idiopathic pulmonary fibrosis
|
OliX Pharmaceuticals, Inc.
|
Preclinical
|
|
OLX-201
|
CTGF
|
siRNA
|
Idiopathic pulmonary fibrosis
|
Korea Institute of Toxicology
|
Preclinical
|
|
Y126S
|
CTGF x PD-1
|
Bispecific antibodies
|
Pancreatic ductal adenocarcinoma
|
Huazhong University of Science and Technology
|
Preclinical
|
|
BLR-200
|
CCN1 x CTGF
|
Synthetic peptides
|
Primary Sclerosing Cholangitis | Novel Coronavirus Infection | interstitial lung disease, etc
|
BLR Bio LLC
|
Preclinical
|
|
RXI-109
|
CTGF
|
siRNA
|
Metastatic breast cancer
|
Phio Pharmaceuticals Corp.
|
Preclinical
|
|
PMB-211
|
CTGF
|
Monoclonal antibodies
|
Fibrosis
|
Protheragen, Inc.
|
Preclinical
|
(Data as of March 13, 2026, source: Synapse)
7. CCN2 Research Tools: Recombinant Proteins, Antibodies and ELISA Kits
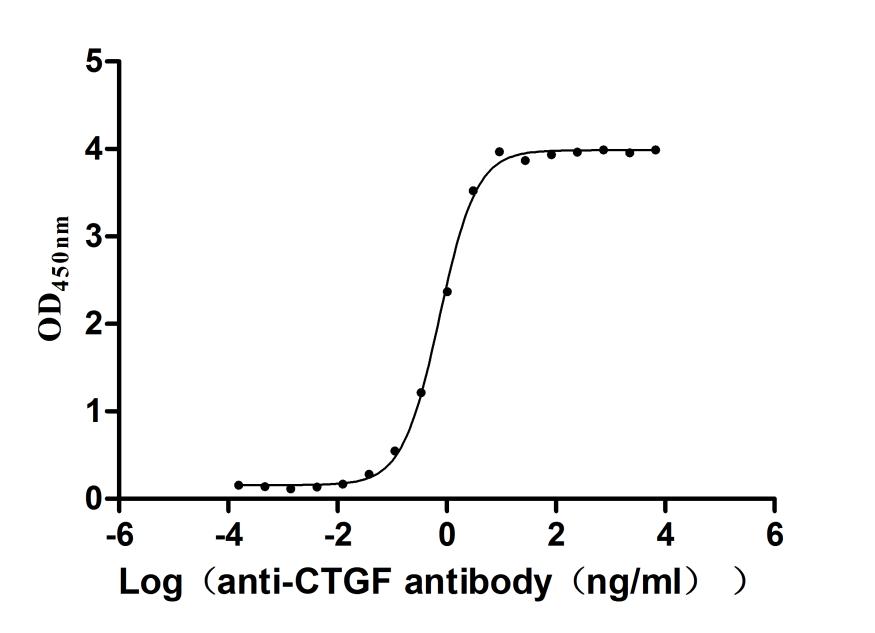
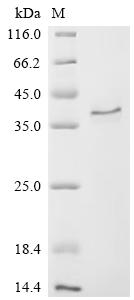
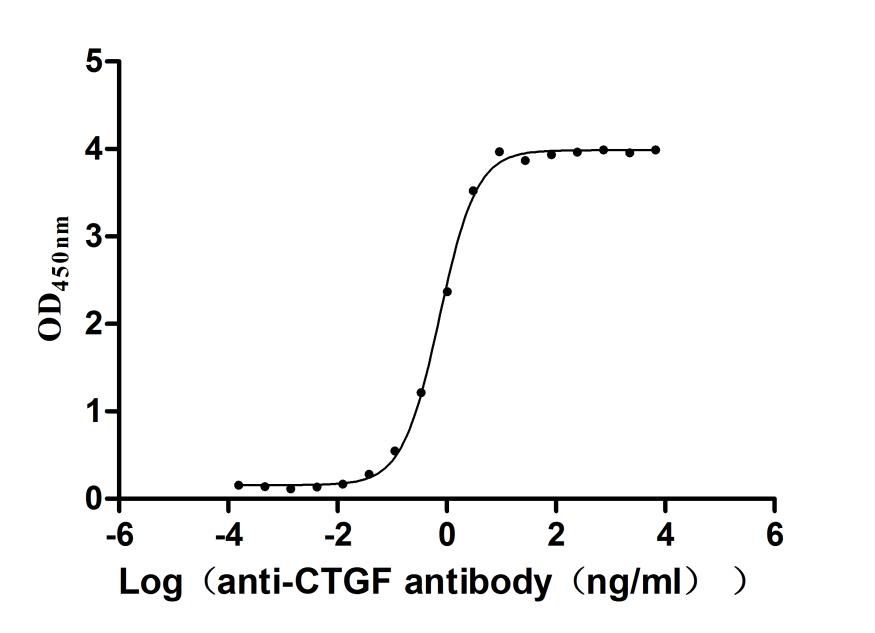
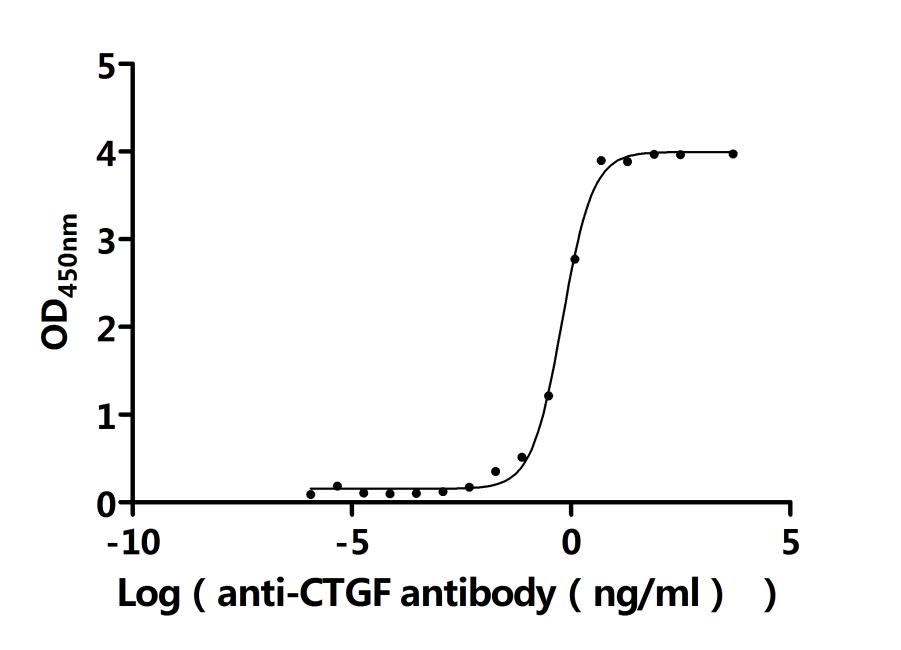
Overall, CCN2 is an important matricellular protein with structural complexity, functional diversity and translational potential, which is closely related to various types of fibrosis, cancer, vascular diseases, metabolic abnormalities and developmental disorders. It is not only an important node in disease mechanism research, but also has potential value as a biomarker and intervention target. Cusabio provides CCN2 recombinant protein, antibody and ELISA kit products to support your mechanism research and targeted drug development.
References
[1] Alexander Peidl, B. Perbal, A. Leask.(2019). Yin/Yang expression of CCN family members: Transforming growth factor beta 1, via ALK5/FAK/MEK, induces CCN1 and CCN2, yet suppresses CCN3, expression in human dermal fibroblasts.
[2] I. Ubink, Elisha R. Verhaar, O. Kranenburg, R. Goldschmeding.(2016). A potential role for CCN2/CTGF in aggressive colorectal cancer.
[3] Priya Ghosh, Ankita Dey, S. Nandi, Ranabir Majumder, Subhayan Das, Mahitosh Mandal.(2025). CTGF (CCN2): a multifaceted mediator in breast cancer progression and therapeutic targeting.
[4] M. Kidd, I. Modlin, G. Eick, R. Camp, S. Mane.(2007). Role of CCN2/CTGF in the proliferation of Mastomys enterochromaffin-like cells and gastric carcinoid development.
[5] O. J. Kaasbøll, A. Gadicherla, Jian-Hua Wang, V. T. Monsen, E. Hagelin, M. Dong, H. Attramadal.(2018). Connective tissue growth factor (CCN2) is a matricellular preproprotein controlled by proteolytic activation.
[6] Hiroaki Amano, Tsutomu Inoue, Takeru Kusano, Daichi Fukaya, Wakako Kosakai, H. Okada.(2023). Module 4-Deficient CCN2/Connective Tissue Growth Factor Attenuates the Progression of Renal Fibrosis via Suppression of Focal Adhesion Kinase Phosphorylation in Tubular Epithelial Cells.
[7] Hannah Murphy-Marshman, Katherine Quensel, X. Shi-wen, R. Barnfield, J. Kelly, Alexander Peidl, R. Stratton, A. Leask.(2017). Antioxidants and NOX1/NOX4 inhibition blocks TGFβ1-induced CCN2 and α-SMA expression in dermal and gingival fibroblasts.
[8] Bram Piersma, Olaf Y. Wouters, Saskia de Rond, M. Boersema, R. A. Gjaltema, R. Bank.(2017). Ascorbic acid promotes a TGFβ1‐induced myofibroblast phenotype switch.
[9] K. Thompson, D. Hamilton, A. Leask.(2010). ALK5 Inhibition Blocks TGFβ-induced CCN2 Expression in Gingival Fibroblasts.
[10] Bin Yu, Guan-nan Jin, Mei Ma, Hui-fang Liang, Bi-xiang Zhang, Xiao-ping Chen, Ze-yang Ding.(2018). Taurocholate Induces Connective Tissue Growth Factor Expression in Hepatocytes Through ERK-YAP Signaling.
[11] X. Liao, Yang Bu, Shanshan Jiang, Fan Chang, Fengan Jia, Xuelian Xiao, Ge Song, Mei Zhang, P. Ning, Qingan Jia.(2019). CCN2–MAPK–Id-1 loop feedback amplification is involved in maintaining stemness in oxaliplatin-resistant hepatocellular carcinoma.
[12] T. Yanagihara, K. Tsubouchi, M. Gholiof, S. Chong, K. Lipson, Q. Zhou, C. Scallan, C. Upagupta, J. Tikkanen, S. Keshavjee, K. Ask, M. Kolb.(2021). Connective-Tissue Growth Factor (CTGF/CCN2) Contributes to TGF-β1-Induced Lung Fibrosis.
[13] Xiaojing Wang, Tianyuan Xu, Fengbin Gao, Hongchao He, Yu Zhu, Zhoujun Shen.(2017). Targeting of CCN2 suppresses tumor progression and improves chemo-sensitivity in urothelial bladder cancer.
[14] C. Chang, M-H Yang, M-H Yang, Been-Ren Lin, S-T Chen, Szu-Hua Pan, M. Hsiao, T-C Lai, S-K Lin, Y. Jeng, C. Chu, R. Chen, P.-C. Yang, P.-C. Yang, Y. E. Chin, M. Kuo.(2012). CCN2 inhibits lung cancer metastasis through promoting DAPK-dependent anoikis and inducing EGFR degradation.
[15] Yu Wang, Xuesong Liu, Qian Xu, Wei Xu, Xianming Zhou, Zhiyong Lin.(2023). CCN2 deficiency in smooth muscle cells triggers cell reprogramming and aggravates aneurysm development.
[16] Yiming Shao, Lei Sun, Baodong Ma, Ranran Jin, Yueyao Ban, Ruibo Li, Jianfa Wang, Hongkai Lian, Han Yue.(2023). VCAM-1 Promotes Angiogenesis of Bone Marrow Mesenchymal Stem Cells Derived from Patients with Trauma-Induced Osteonecrosis of the Femoral Head by Regulating the Apelin/CCN2 Pathway.
[17] Zhifei Xu, Yixiao Jin, Zizheng Gao, Yan Zeng, Jiangxia Du, Hao Yan, Xueqin Chen, Li Ping, Nengming Lin, Bo Yang, Qiaojun He, P. Luo.(2021). Autophagic degradation of CCN2 (cellular communication network factor 2) causes cardiotoxicity of sunitinib.
CUSABIO team. CCN2 (CTGF) Target: Core Molecule of Fibrosis and Tumor Stroma Regulation | Disease Treatment Research Progress. https://www.cusabio.com/c-21303.html



Comments
Leave a Comment