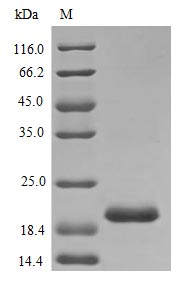
Prolactin drives JAK2/STAT5 signaling through the prolactin receptor, making validated bioactivity essential for meaningful downstream results. This tag-free recombinant rat Prl, spanning residues 30–226 of the mature protein, demonstrates an ED50 below 1.0 ng/ml in Nb2-11 cell proliferation assays—corresponding to a specific activity exceeding 1.0 × 10⁶ IU/mg—confirming its suitability for receptor binding assays, STAT5 phosphorylation studies, and cell-based signaling experiments such as cAMP induction. Endotoxin levels below 1.0 EU/μg, combined with purity greater than 98% by SDS-PAGE, satisfy the criteria typically required for in vivo pharmacology models and antibody development programs where contaminating signals must be minimized. Researchers can confidently employ this protein as an immunoassay calibration standard or in structure-function studies exploring prolactin receptor engagement.