4. The Application of Serum Albumin
Serum albumin is an important biological product, which is widely used in clinic and biotechnology.
Biomarkers
Serum albumin is a valuable biomarker of many diseases, including cancer, rheumatoid arthritis, ischemia, post-menopausal obesity, severe acute graft-versus-host disease, and diseases that need monitoring of the glycemic control.
Hypoalbuminemia often occurs in patients with active rheumatoid arthritis, mainly caused by high HSA uptake in inflammatory sites. As a biomarker of blood glucose control during pregnancy, glycosylated serum albumin reflects the short-term state of blood glucose control and is considered as a good biomarker of blood glucose control.
Determination of serum albumin level in blood is one of the methods to evaluate the nutritional status, severity and progression of cancer patients. Serum albumin has also been described as an independent prognosticator of survival in various cancers, such as lung cancer[15], pancreas[16], gastric cancer[17], colorectal cancer[18] and breast cancer[19].
Clinical Application
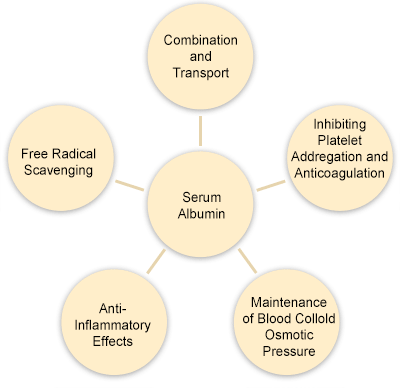
Albumin can be widely used as a plasma volume extender in many clinical conditions, such as hypovolemic, shock, burn, surgery, trauma, acute respiratory distress syndrome, hemodialysis, acute liver failure, hypoalbuminosis, etc[20]. Albumin also plays a role in protecting ischemic and hypoxic liver injury, mainly due to its strong antioxidant characteristics.
As a carrier of drugs, serum albumin reversibly binds to most drugs in the blood circulation. The binding with serum albumin has great influence on the pharmacokinetics and pharmacodynamics of most commonly used prescription drugs. For example, binding to serum albumin increases the solubility of lipophilic drugs, allowing them to reach the site of action. However, conjugated drugs cannot easily leave the capillary, only unbound drugs can be distributed into tissues, thus exerting pharmacological activity or toxicity. Therefore, the high affinity between drugs and serum albumin may be beneficial or detrimental to its efficacy. In addition to affecting the distribution of drugs, binding to serum albumin also affects drug metabolism and elimination.
Biotechnology Aspects
Recently, biotechnological applications of HSA, including implantable biomaterials, surgical adhesives and sealants, biochromatography, ligand trapping, and fusion proteins, have been reported. HSA is also used as an excipient for vaccines or therapeutic protein drugs and as a cell culture medium supplement in the production of vaccines and pharmaceuticals[21]. Moreover, HSA could act as a carrier of O2[22], drugs[23], and fusion peptides[24].
Supplements: As a supplement to cell growth and productivity, serum albumin is added to the cell culture medium[25]. As serum albumin has the ability to carry and deliver important nutrients to cells, it can improve the overall health of cells. Serum albumin can also bind free radicals to reduce damage to cells.
HSA nanotubes: Recently, HSA nanotubes trapping either HSA binding molecules or biotin or hepatitis B virus and displaying catalytic activity have been obtained.
Remove bilirubin: Serum albumin can be used to remove bilirubin during dialysis. What we know about bilirubin is that a significant increase in plasma bilirubin levels may lead to severe neurological damage, especially in newborns. Therefore, regulating the cell content of bilirubin by binding, oxidation and output is essential to cell health.
Serum albumin fusion protein: The fusion of serum albumin and peptide with therapeutic function has also been used effectively. Furukawa et al. have produced fusion proteins of human serum albumin and thioredoxin (HSA-TRX), HSA TRX fusion protein has proven to be therapeutically effective in the septic shock mouse model[24]. Using the characteristic of HSA accumulation in tumor and long half-life, it can be used for tumor localization by binding it to tumor monoclonal antibody.
References:
[1] Ahn S M, Byun K, Cho K, et al. Human microglial cells synthesize albumin in brain [J]. Plos One, 2008, 3(7): e2829.
[2] Yamaguchi M, Igarashi A, Misawa H, et al. Enhancement of albumin expression in bone tissues with healing rat fractures [J]. Journal of Cellular Biochemistry, 2010, 89(2).
[3] Mendez C M, Mcclain C J, Marsano L S. Albumin therapy in clinical practice [J]. Nutrition in Clinical Practice Official Publication of the American Society for Parenteral & Enteral Nutrition, 2005, 20(3): 314.
[4] Nicholson J P, Wolmarans M R, Park G R. The role of albumin in critical illness [J]. British Journal of Anaesthesia, 2000, 85(4): 599-610.
[5] Quinlan G J, Martin G S, Evans T W. Albumin: biochemical properties and therapeutic potential [J]. Hepatology, 2010, 41(6): 1211-9.
[6] Bolli A, Marino M, Rimbach G, et al. Flavonoid binding to human serum albumin [J]. Biochemical & Biophysical Research Communications, 2010, 398(3): 444-449.
[7] Curry S. Lessons from the crystallographic analysis of small molecule binding to human serum albumin [J]. Drug Metabolism & Pharmacokinetics, 2009, 24(4): 342-357.
[8] Vusse G J V D. Albumin as Fatty Acid Transporter [J]. Drug Metabolism & Pharmacokinetics, 2009, 24(4): 300-307.
[9] Fasano M, Curry S, Terreno E, et al. The extraordinary ligand binding properties of human serum albumin [J]. Iubmb Life, 2010, 57(12): 787-796.
[10] Ascenzi P, Bocedi A, Notari S, et al. Allosteric modulation of drug binding to human serum albumin [J]. Mini Reviews in Medicinal Chemistry, 2006, 6(4).
[11] Abe T, Abe T, Ageta S, et al. A new method for removal of albumin-binding uremic toxins: efficacy of an albumin-dialysate [J]. Therapeutic Apheresis Official Journal of the International Society for Apheresis & the Japanese Society for Apheresis, 2010, 5(1): 58-63.
[12] Pedersen T, Møller A M, Gøtzsche P C. Human albumin in critically ill patients [J]. Critical Care Medicine, 2005, 33(5): 1183-5.
[13] Strubelt O, Younes M, Li Y. Protection by Albumin against Ischaemia and Hypoxia Induced Hepatic Injury [J]. Basic & Clinical Pharmacology & Toxicology, 2010, 75(3-4): 280-284.
[14] Chien S C, Chen C Y, Lin C F, et al. Critical appraisal of the role of serum albumin in cardiovascular disease [J]. Biomarker Research, 2017, 5(1): 31.
[15] Lam P T, Leung M W, Tse C Y. Identifying prognostic factors for survival in advanced cancer patients: a prospective study [J]. Hong Kong Med J, 2007, 13(6): 453-459.
[16] Siddiqui 25 Siddiqui A, Heinzerling J, Livingston E H, et al. Predictors of early mortality in veteran patients with pancreatic cancer [J]. American Journal of Surgery, 2007, 194(3): 362-366.
[17] Oñateocaña L F, Aiellocrocifoglio V, Gallardorincón D, et al. Serum Albumin as a Significant Prognostic Factor for Patients with Gastric Carcinoma [J]. Annals of Surgical Oncology, 2007, 14(2): 381-389.
[18] Cengiz O, Kocer B, Sürmeli S, et al. Are pretreatment serum albumin and cholesterol levels prognostic tools in patients with colorectal carcinoma? [J]. Medical Science Monitor International Medical Journal of Experimental & Clinical Research, 2006, 12(6): CR240.
[19] Lis C G, Grutsch J F, Vashi P G, et al. Is serum albumin an independent predictor of survival in patients with breast cancer? [J]. Jpen J Parenter Enteral Nutr, 2003, 27(1): 10-15.
[20] Aramwit P, Kasettratat N. Evaluation of serum albumin utilization in inpatient at a private hospital in Bangkok [J]. Yakugaku Zasshi Journal of the Pharmaceutical Society of Japan, 2004, 124(9): 631.
[21] Marth E, Kleinhappl B. Albumin is a necessary stabilizer of TBE-vaccine to avoid fever in children after vaccination [J]. Vaccine, 2001, 20(3): 532-537.
[22] Tsuchida E, Sou K, Nakagawa A, et al. Artificial oxygen carriers, hemoglobin vesicles and albumin-hemes, based on bioconjugate chemistry [J]. Bioconjugate Chemistry, 2009, 20(8): 1419.
[23] Cai C, Zhou K, Wu Y, et al. Enhanced liver targeting of 5-fluorouracil using galactosylated human serum albumin as a carrier molecule [J]. Journal of Drug Targeting, 2006, 14(2): 55-61.
[24] Furukawa M, Tanaka R, Chuang V T, et al. Human serum albumin-thioredoxin fusion protein with long blood retention property is effective in suppressing lung injury [J]. Journal of Controlled Release Official Journal of the Controlled Release Society, 2011, 154(2): 189-195.
[25] Fanali G, Di M A, Trezza V, et al. Human serum albumin: from bench to bedside [J]. Molecular Aspects of Medicine, 2012, 33(3): 209-290.
[26] Yang D, Guo F, Liu B, et al. Expression and localization of human lysozyme in the endosperm of transgenic rice [J]. Planta, 2003, 216(4): 597-603.
[27] Huang N, Bethell D, Card C, et al. Bioactive recombinant human lactoferrin, derived from rice, stimulates mammalian cell growth [J]. In Vitro Cellular & Developmental Biology. Animal, 2008, 44(10): 464-471.
[28] He Y, Ning T, Xie T, et al. Large-scale production of functional human serum albumin from transgenic rice seeds [J]. Proceedings of the National Academy of Sciences of the United States of America, 2011, 108(47): 19078-19083.



Comments
Leave a Comment