[1] Kayoko Hase, K. Tani, Teruki Shimizu, Y. Ohmoto, K. Matsushima, S. Sone.(2001). Increased CCR4 expression in active systemic lupus erythematosus.
[2] Kenji Ishitsuka, Tomoharu Yasukawa, Yukie Tsuji.(2024). Safety and effectiveness of mogamulizumab in relapsed or refractory CC chemokine receptor 4‐positive peripheral T‐cell lymphoma and relapsed or refractory cutaneous T‐cell lymphoma: A post‐marketing surveillance in Japan.
[3] Xi Cheng, Huo Wu, Zhi-Jian Jin, Ding Ma, Stanley Yuen, Xiaoqian Jing, Minmin Shi, B. Shen, C. Peng, R. Zhao, Wei-hua Qiu.(2017). Up-regulation of chemokine receptor CCR4 is associated with Human Hepatocellular Carcinoma malignant behavior.
[4] Y. Yamano, N. Araya, Tomoo Sato, A. Utsunomiya, Kazuko Azakami, D. Hasegawa, Toshihiko Izumi, Hidetoshi Fujita, S. Aratani, N. Yagishita, Ryoji Fujii, K. Nishioka, S. Jacobson, T. Nakajima.(2009). Abnormally High Levels of Virus-Infected IFN-γ+CCR4+CD4+CD25+ T Cells in a Retrovirus-Associated Neuroinflammatory Disorder.
[5] C. Carola, Andrew Salazar, C. Rakers, F. Himbert, Q. Do, P. Bernard, Joerg von Hagen.(2021). A Cornflower Extract Containing N-Feruloylserotonin Reduces Inflammation in Human Skin by Neutralizing CCL17 and CCL22 and Inhibiting COX-2 and 5-LOX.
[6] Yongjian Dong, Shuhui Gao, Xue-fang Zhang, Jing Kou, Jing Liu, Ting Ye, Han Shen.(2021). CCL17 and CCL22 induce CCR4 receptor expression and promote cytokine-induced killer cells migration.
[7] Inés Lecoq, K. Kopp, M. Chapellier, Panagiotis Mantas, E. Martinenaite, Maria Perez-Penco, Lars Rønn Olsen, M. Zocca, A. Wakatsuki Pedersen, M. Andersen.(2022). CCL22-based peptide vaccines induce anti-cancer immunity by modulating tumor microenvironment.
[8] Chun Jin, Liangliang Lu, Jian Gao, Ling Chen.(2024). M2-like Macrophages-derived CCL17 Promotes Esophageal Squamous Cell Carcinoma Metastasis and Stemness via Activating CCR4-mediated ERK/PD-L1 Pathway..
[9] A. Maolake, K. Izumi, K. Shigehara, Ariunbold Natsagdorj, H. Iwamoto, S. Kadomoto, Yuta Takezawa, Kazuaki Machioka, K. Narimoto, M. Namiki, Wen-Jye Lin, Guzailinuer Wufuer, A. Mizokami.(2016). Tumor-associated macrophages promote prostate cancer migration through activation of the CCL22–CCR4 axis.
[10] W. Gan, Baoye Sun, Zhangfu Yang, Cheng Ye, Zhutao Wang, Cheng Zhou, Guo-Qiang Sun, Yong Yi, Shuang-Jian Qiu.(2024). Enhancing hepatocellular carcinoma management: prognostic value of integrated CCL17, CCR4, CD73, and HHLA2 expression analysis.
[11] Lichao Zhang, Xiaoqing Lu, Yuanzhi Xu, Xiaoqin La, Jinmiao Tian, Aiping Li, Hanqing Li, Changxin Wu, Yanfeng Xi, Guisheng Song, Zhaocai Zhou, Wenqi Bai, Liwei An, Zhuoyu Li.(2023). Tumor-associated macrophages confer colorectal cancer 5-fluorouracil resistance by promoting MRP1 membrane translocation via an intercellular CXCL17/CXCL22–CCR4–ATF6–GRP78 axis.
[12] Shuixiang Deng, Peng Jin, P. Sherchan, Shengpeng Liu, Yuhui Cui, Lei Huang, John H. Zhang, Ye Gong, Jiping Tang.(2021). Recombinant CCL17-dependent CCR4 activation alleviates neuroinflammation and neuronal apoptosis through the PI3K/AKT/Foxo1 signaling pathway after ICH in mice.
[13] Rui Lin, Y. Choi, David A Zidar, Julia K. L. Walker.(2018). β‐Arrestin‐2‐Dependent Signaling Promotes CCR4‐mediated Chemotaxis of Murine T‐Helper Type 2 Cells.
[14] H. Lim, J. R. Lane, Meritxell Canals, M. Stone.(2021). Systematic Assessment of Chemokine Signaling at Chemokine Receptors CCR4, CCR7 and CCR10.
[15] Tania Sarkar, Subhanki Dhar, D. Chakraborty, Subhadip Pati, S. Bose, A. Panda, Udit Basak, S. Chakraborty, S. Mukherjee, A. Guin, Kuladip Jana, Diptendra K. Sarkar, Gaurisankar Sa.(2022). FOXP3/HAT1 Axis Controls Treg Infiltration in the Tumor Microenvironment by Inducing CCR4 Expression in Breast Cancer.
[16] Li Zhang, Sijuan Tian, Jie Chang, Shimin Quan, Ting Yang, Minyi Zhao, Li Wang, Xiaofeng Yang.(2024). Activation of the CCL22/CCR4 causing EMT process remodeling under EZH2-mediated epigenetic regulation in cervical carcinoma.
[17] Y. Li, J. Srinivasan, Hilary J. Selden, Lauren I. R. Ehrlich.(2023). CCR4 and CCR7 differentially regulate thymocyte localization with distinct outcomes for central tolerance.
[18] Z. Marušić, E. Calonje.(2021). Skin adnexal tumours in a tertiary dermatopathology service.
[19] Do-Youn Oh, Min-Hee Ryu, Jun-Eul Hwang, Jaeyong Cho, D. Y. Zang, Sang Cheul Oh, J. Lee, Keun-Wook Lee, S. Rha, B. Shim, William Ho, M. Chisamore, Paul Dong Rhee, Sooa Jung, Taewan Kim, Jiyeon Yoon, Hosung Jang, E. Baek, Young-Su Noh.(2025). A phase 2 study to assess the safety and efficacy of FLX475 (tivumecirnon) combined with pembrolizumab in patients with advanced or metastatic gastric cancer..
[20] Adam Grant, Juraj Adamik, Jingtao Qiu, Jacob Wert, Molly Grandcolas, S. Jacobson, R. Goyal, William Ho, Shoji Ikeda, Marvin Au, Damian L Trujillo, M. Chisamore, D. Nagata, M. S. Ghomi, D. Brockstedt, P. Kassner, George E. Katibah.(2024). Abstract 2485: A combined mregDC and Treg signature associates with antitumor efficacy of CCR4 antagonist tivumecirnon FLX475.
[21] Y. Mahnke, K. Fletez-Brant, I. Sereti, M. Roederer.(2016). Reconstitution of Peripheral T Cells by Tissue-Derived CCR4+ Central Memory Cells Following HIV-1 Antiretroviral Therapy.
[22] J. Bogacka, Katarzyna Ciapała, K. Pawlik, K. Kwiatkowski, J. Dobrogowski, A. Przeklasa-Muszyńska, J. Mika.(2020). CCR4 Antagonist (C021) Administration Diminishes Hypersensitivity and Enhances the Analgesic Potency of Morphine and Buprenorphine in a Mouse Model of Neuropathic Pain.

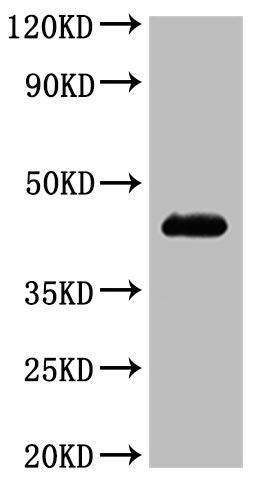
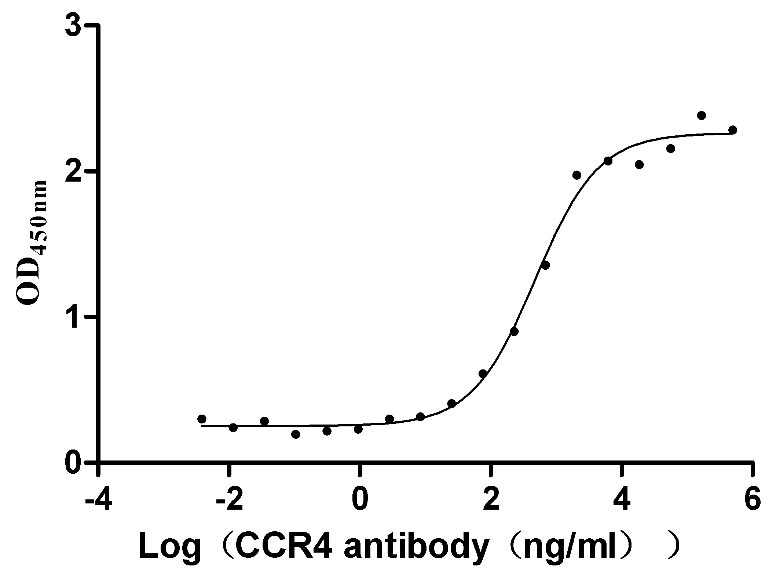
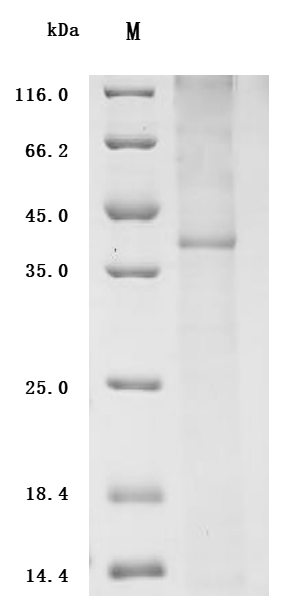
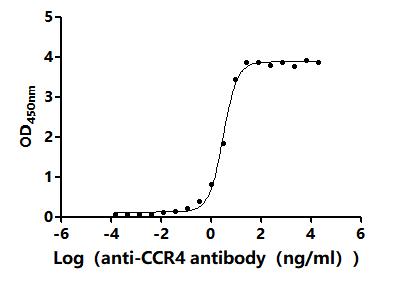
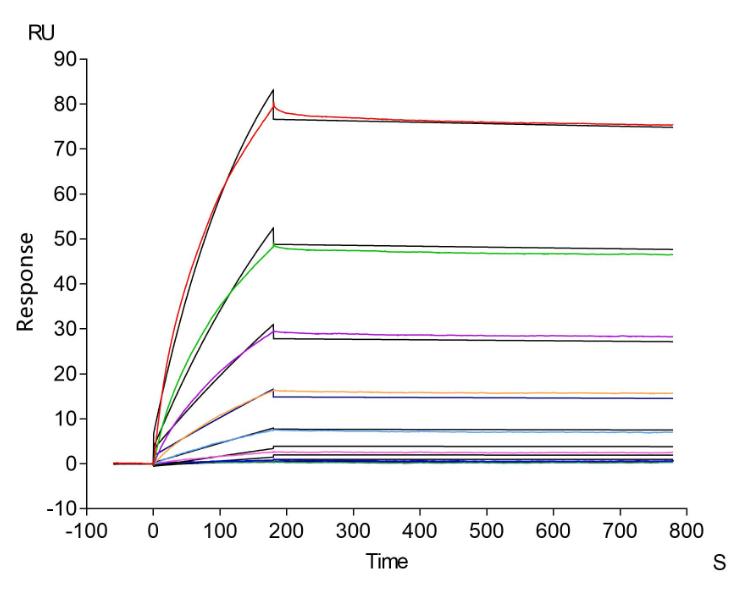
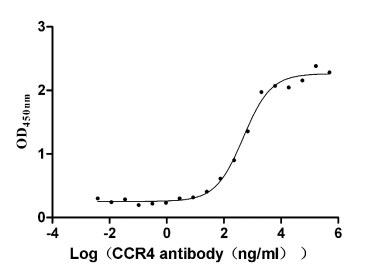
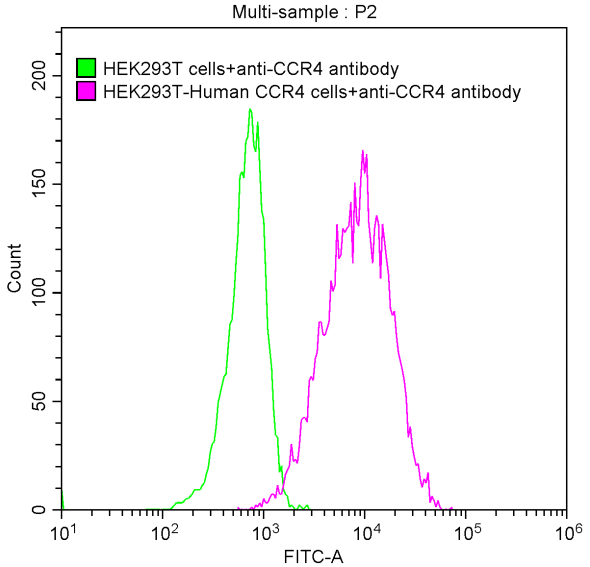


Comments
Leave a Comment