[1] S. Sarabipour, K. Hristova. (2016). Mechanism of FGF receptor dimerization and activation.
[2] Yuga Okada, Akihiro Eguchi, Daisuke Kuroda, Kouhei Tsumoto, Ryosuke Ueki, Shinsuke Sando. (2025). Developing FGF2 Mutants with Selectively Reduced Heparan Sulfate Affinity to Explore Their Impact on FGFR1 Signaling.
[3] W. Chan, K. Howe, James M. Clegg, S. Guimond, D. Price, J. Turnbull, T. Pratt. (2015). 2-O Heparan Sulfate Sulfation by Hs2st Is Required for Erk/Mapk Signalling Activation at the Mid-Gestational Mouse Telencephalic Midline.
[4] Dasol Han, S. Lee, Mookwang Kwon, Hogyun Noh, J. H. Lee, Y. Yoon, J. Cho, K. Yoon. (2020). YAP enhances FGF2-dependent neural stem cell proliferation by induction of fibroblast growth factor receptor expression.
[5] Sebastián Giulianelli, Marina Riggio, Tomás Guillardoy, C. Pérez Piñero, M. Gorostiaga, G. Sequeira, Gabriela Pataccini, M. F. Abascal, Maria F. Toledo, B. Jacobsen, Ana C. L. Guerreiro, António S. Barros, V. Novaro, F. Monteiro, Francisco Amado, H. Gass, M. Abba, L. Helguero, C. Lanari. (2019). FGF2 induces breast cancer growth through ligand‑independent activation and recruitment of ERα and PRBΔ4 isoform to MYC regulatory sequences.
[6] Bin Zeng, Guosheng Lin, X. Ren, Yan Zhang, Honglei Chen. (2010). Over-expression of HO-1 on mesenchymal stem cells promotes angiogenesis and improves myocardial function in infarcted myocardium.
[7] Xiaotang Wang, W. Fan, Na Li, Yan Ma, Mudi Yao, Guoqing Wang, Siyuan He, Wanqian Li, J. Tan, Qingjun Lu, Shengping Hou. (2021). YY1 lactylation in microglia promotes angiogenesis through transcription activation-mediated upregulation of FGF2.
[8] P. Sun, Feifei Ma, Yang Xu, Chao Zhou, R. A. Stetler, K. Yin. (2021). Genetic deletion of endothelial microRNA-15a/16-1 promotes cerebral angiogenesis and neurological recovery in ischemic stroke through Src signaling pathway.
[9] Lushu Chen, Yuan Cao, Yaming Shen, Huan Li, Rong Ye, Jin Yao. (2023). Downregulation of PIK3IP1 in retinal microglia promotes retinal pathological neovascularization via PI3K-AKT pathway activation.
[10] A. Fouda, Zhimin Xu, J. Suwanpradid, M. Rojas, E. Shosha, Tahira Lemtalsi, C. Patel, Ji Xing, Sumbal Zaidi, W. Zhi, Brain K Stansfield, P. Cheng, S. Narayanan, R. Caldwell, R. Caldwell. (2022). Targeting proliferative retinopathy: Arginase 1 limits vitreoretinal neovascularization and promotes angiogenic repair.
[11] Zijie Wang, Chuying Chen, Liying Sun, Mei He, Ting Huang, Jiji Zheng, Jiayuan Wu. (2024). Fibroblast growth factor 2 promotes osteo/odontogenic differentiation in stem cells from the apical papilla by inhibiting PI3K/AKT pathway.
[12] Zhonghao Wang, Yexin Wang, Yu Tang, Xiaoyan Guo, Qize Gao, Yiming Shao, Jingxuan Wang, Ronghua Tian, Yingxu Shi. (2025). Sodium Benzoate Inhibits Osteoblast Differentiation and Accelerates Bone Loss by Regulating the FGF2/p38/RUNX2 Pathway.
[13] Ok-Jin Park, Hyun-Jung Kim, K. Woo, J. Baek, H. Ryoo. (2009). FGF2-activated ERK Mitogen-activated Protein Kinase Enhances Runx2 Acetylation and Stabilization.
[14] Yang Jiao, Jing Xu, Bingtan Song, Ailing Wu, L. Pan, Ying Xu, Fenghao Geng, Xiaoqian Li, Congzhao Zhao, Min Hong, Xuanyu Meng, Judong Luo, Peng Liu, Ming Li, Wei Zhu, Jianping Cao, Shuyu Zhang. (2022). Interferon regulatory factor 1‐triggered free ubiquitin protects the intestines against radiation‐induced injury via CXCR4/FGF2 signaling.
[15] Mengwei Wang, Yuanmeng Sun, Yanzhi Zhao, Xinyi Jiang, Teng Wang, Junye Xie, Xiuling Yu, Shujun Guo, Yibo Zhang, Xiaojia Chen, An Hong. (2025). An FGF2-Derived Short Peptide Attenuates Bleomycin-Induced Pulmonary Fibrosis by Inhibiting Collagen Deposition and Epithelial--Mesenchymal Transition via the FGFR/MAPK Signaling Pathway.
[16] R. Harris, A. Adeyemo, J. Redfield, Cha-hua Huang, Min Jiang, Xiaoping Ren, W. Jones, Jack Rubinstein, J. J. Schultz. (2016). Long‐term Biological Functions of Fibroblast Growth Factor‐2 (FGF2) in Mouse Hearts Following Myocardial Infarction.
[17] Josh W DiGiacomo, Inês Godet, Michael Trautmann-Rodriguez, Daniele M. Gilkes. (2020). Extracellular Matrix--Bound FGF2 Mediates Estrogen Receptor Signaling and Therapeutic Response in Breast Cancer.
[18] P. Têtu, J. Delyon, J. André, Coralie Reger de Moura, Malak Sabbah, G. Ghanem, M. Battistella, S. Mourah, C. Lebbé, N. Dumaz. (2020). FGF2 Induces Resistance to Nilotinib through MAPK Pathway Activation in KIT Mutated Melanoma.
[19] Yu Zhang, Yan Wang, Di Zhou, Kai Wang, Xu Wang, Xiang Wang, Yang Jiang, Min Zhao, Rutong Yu, Xiuping Zhou. (2021). Radiation-induced YAP activation confers glioma radioresistance via promoting FGF2 transcription and DNA damage repair.
[20] Iwona Bednarz-Misa, Paulina Fortuna, D. Diakowska, Natalia Jamrozik, M. Krzystek-Korpacka. (2020). Distinct Local and Systemic Molecular Signatures in the Esophageal and Gastric Cancers: Possible Therapy Targets and Biomarkers for Gastric Cancer.
[21] Z. Alayash, S. Baumeister, B. Holtfreter, Thomas Kocher, H. Baurecht, B. Ehmke, D. Hagenfeld, S. L. Reckelkamm, M. Nolde. (2025). Proteome-Guided Drug Target Discovery for Periodontitis.

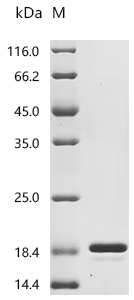
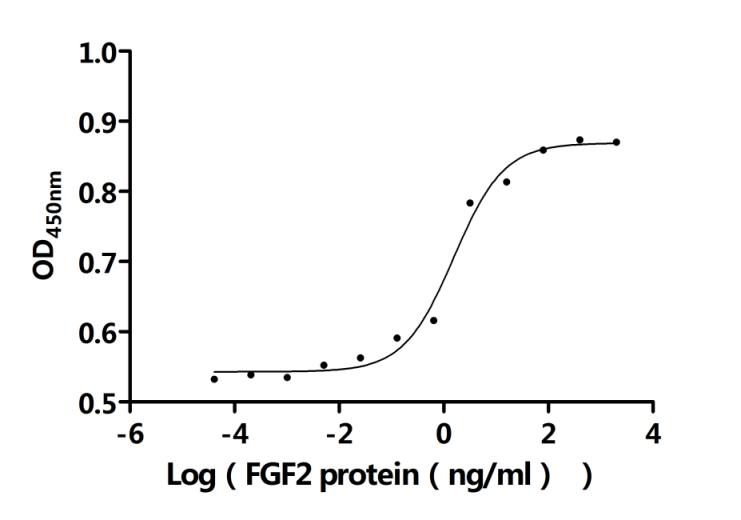
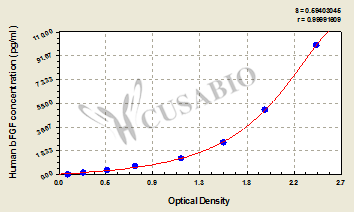
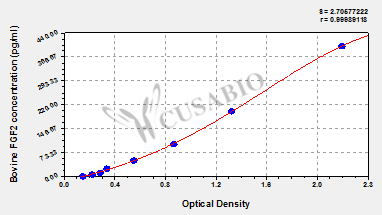


Comments
Leave a Comment