[1] L. Romero-Castillo, Taotao Li, Nhu-Nguyen Do, O. Sareila, Bingze Xu, Viktoria Hennings, Zhongwei Xu, Carolin Svensson, Ana Oliveira-Coelho, Zeynep Sener, V. Urbonaviciute, O. Ekwall, Harald Burkhardt, Rikard Holmdahl. (2024). Human MHC Class II and Invariant Chain Knock‐in Mice Mimic Rheumatoid Arthritis with Allele Restriction in Immune Response and Arthritis Association.
[2] Lin Zhang, Iris Woltering, Mathias Holzner, Markus Brandhofer, Carl-Christian Schaefer, Genta Bushati, Simon Ebert, Bishan Yang, Maximilian Muenchhoff, J. Hellmuth, C. Scherer, Christian Wichmann, David Effinger, Max Hübner, O. El Bounkari, Patrick Scheiermann, J. Bernhagen, A. Hoffmann. (2024). CD74 is a functional MIF receptor on activated CD4+ T cells.
[3] H. Bergmann. (2012). Business as usual: the p35 isoform of human CD74 retains function in antigen presentation.
[4] Shuchun Zhao, A. Molina, Abigail S. Yu, J. Hanson, Harry Cheung, Xiaofan Li, Y. Natkunam. (2018). High frequency of CD74 expression in lymphomas: implications for targeted therapy using a novel anti‐CD74-drug conjugate.
[5] C. Abrahams, Xiaofan Li, M. Embry, Abigail S. Yu, Stellanie Krimm, S. Krueger, Nancy Y. Greenland, K. Wen, C. Jones, V. Dealmeida, W. Solis, S. Matheny, T. Kline, A. Yam, R. Stafford, Arun P. Wiita, T. Hallam, M. Lupher, A. Molina. (2018). Targeting CD74 in multiple myeloma with the novel, site-specific antibody-drug conjugate STRO-001.
[6] Xiaofan Li, C. Abrahams, Abigail S. Yu, M. Embry, R. Henningsen, V. Dealmeida, S. Matheny, T. Kline, A. Yam, R. Stafford, T. Hallam, M. Lupher, Arturo Molina. (2023). Targeting CD74 in B-cell non-Hodgkin lymphoma with the antibody-drug conjugate STRO-001.
[7] Guosheng Wu, Yu Sun, Kangan Wang, Zhengli Chen, Xingtong Wang, Fei Chang, Ting Li, P. Feng, Z. Xia. (2016). Relationship between elevated soluble CD74 and severity of experimental and clinical ALI/ARDS.
[8] Rossin Erbe, M. Stein, Tim A. Rand, J. Guinney. (2024). Abstract 2281: A tumor-intrinsic signature involving immunosuppression via MIF-CD74 signaling is associated with overall survival in ICT-treated lung adenocarcinoma.
[9] Mona M.Hosseini, Trevor Enright, Adam S. Duvall, Alex Chitsazan, Hsin-Yun Lin, A. Ors, B. Davis, Olga Nikolova, Erica Bresciani, J. Diemer, K. Craft, Catarina Menezes, M. Merguerian, Shawn Chong, Katherine R. Calvo, Shira G Glushakow-Smith, K. Gritsman, Lucy Godley, M. Horwitz, Sioban Keel, Lucio H Castilla, Emek Demir, H. Mohammed, Paul Liu, A. Agarwal. (2024). CD74 Signaling Regulates Inflammatory Stress to Drive Defective Hematopoiesis and Fitness Advantage of Progenitors in RUNX1-Familial Platelet Disorder.
[10] J. Zhou, Alice Shasha Cheng, Li Chen, L. X. Li, Ewud Agborbesong, Vicente E Torres, Peter C. Harris, Xiaogang Li. (2024). CD74 Promotes Cyst Growth and Renal Fibrosis in Autosomal Dominant Polycystic Kidney Disease.
[11] Sarah Zerimech, Hung Nguyen, A. Vandenbark, H. Offner, S. Baltan. (2023). Novel therapeutic for multiple sclerosis protects white matter function in EAE mouse model.
[12] Y. Zhao, Y. Tu, Q. Yang, Y. Zhang, D. Zou. (2025). OP21 MIF-CD74 signaling promotes intestinal LTi-like ILC3s activation and gut fibrosis via upregulating pSTAT3.
[13] Lisa Werr, Dennis Plenker, M. A. Dammert, C. Lorenz, J. Brägelmann, H. Tumbrink, Sebastian Klein, A. Schmitt, R. Büttner, T. Persigehl, K. Shokat, F. Wunderlich, A. Schram, M. Peifer, M. Sos, H. Reinhardt, Roman K. Thomas. (2022). CD74-NRG1 Fusions Are Oncogenic In Vivo and Induce Therapeutically Tractable ERBB2:ERBB3 Heterodimerization.
[14] Woosol Chris Hong, D. Lee, H. Kang, Myeong Jin Kim, Minsoo Kim, J. Kim, S. Fang, H. Kim, Joon Seong Park. (2023). CD74 Promotes a Pro-Inflammatory Tumor Microenvironment by Inducing S100A8 and S100A9 Secretion in Pancreatic Cancer.
[15] J. Burton, S. Ely, P. K. Reddy, R. Stein, D. Gold, Thomas M Cardillo, D. Goldenberg. (2004). CD74 Is Expressed by Multiple Myeloma and Is a Promising Target for Therapy.
[16] Zixiang Wang, Bingyu Wang, Yuan Feng, Jinwen Ye, Zhonghao Mao, Teng Zhang, Meining Xu, Wenjing Zhang, Xinlin Jiao, Qing Zhang, Youzhong Zhang, Baoxia Cui. (2024). Targeting tumor-associated macrophage-derived CD74 improves efficacy of neoadjuvant chemotherapy in combination with PD-1 blockade for cervical cancer.
[17] Jianghua Wang, Xiaoting Li, Guanxi Xiao, Jayesh Desai, S. Frentzas, Z. Wang, Yu Xia, Baiyong Li. (2024). CD74 is associated with inflamed tumor immune microenvironment and predicts responsiveness to PD-1/CTLA-4 bispecific antibody in patients with solid tumors.
[18] A. Ghoochani, M. Schwarz, E. Yakubov, T. Engelhorn, A. Doerfler, M. Buchfelder, R. Bucala, N. Savaskan, I. Eyüpoglu. (2016). MIF-CD74 signaling impedes microglial M1 polarization and facilitates brain tumorigenesis.
[19] R. Meza‐Romero, G. Benedek, Grant Gerstner, G. Kent, Ha Nguyen, H. Offner, A. Vandenbark. (2018). Increased CD74 binding and EAE treatment efficacy of a modified DRα1 molecular construct.
[20] Jiangyi Liu, Xingyun Liao, Na Li, Zongren Xu, Wang Yang, Hongxiu Zhou, Yusen Liu, Zhi Zhang, Guoqing Wang, Shengping Hou. (2024). Single‐cell RNA sequencing reveals inflammatory retinal microglia in experimental autoimmune uveitis.
[21] R. Wilbrink, A. Spoorenberg, F. Kroese, G. Verstappen. (2023). POS0436 INCREASED EXPRESSION OF PATHWAYS INVOLVED IN B CELL ACTIVATION IN PATIENTS WITH ANKYLOSING SPONDYLITIS.

-SDS.jpg)
-AC1.jpg)
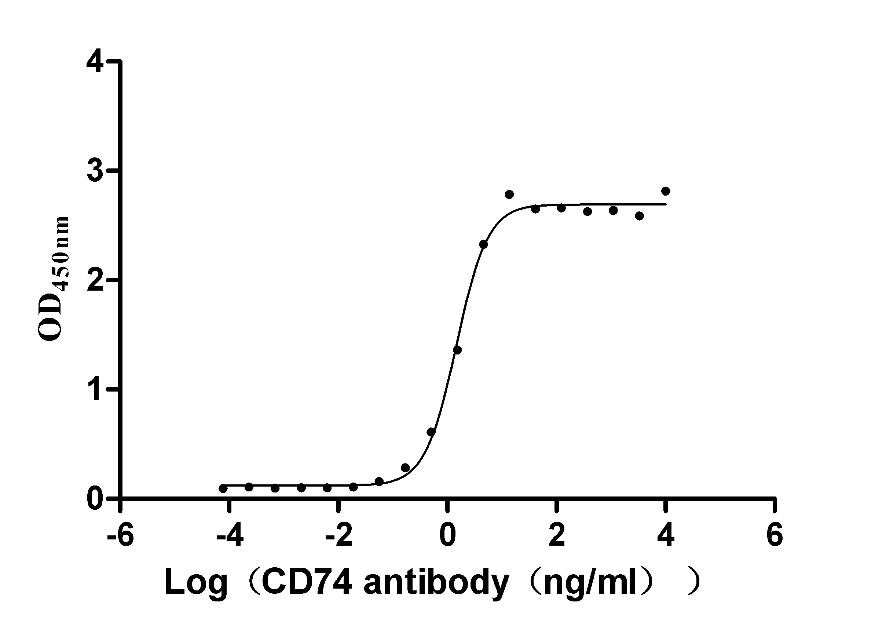
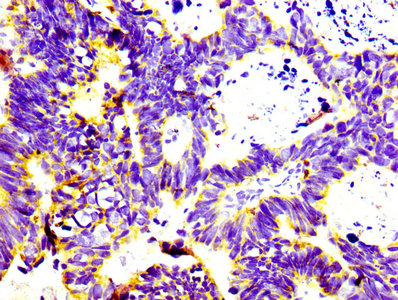
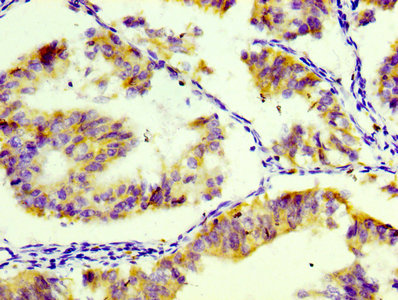
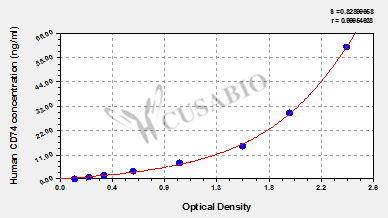


Comments
Leave a Comment