This recombinant mouse Mapt protein (amino acids 2-733) represents the full-length mature form of this crucial neuronal protein, expressed in mammalian cells with an N-terminal 10×His tag. The recombinant Mapt protein demonstrates high purity (>90% by SDS-PAGE) and minimal endotoxin contamination (<1.0 EU/μg, LAL method), making it suitable for sensitive neurological research applications.
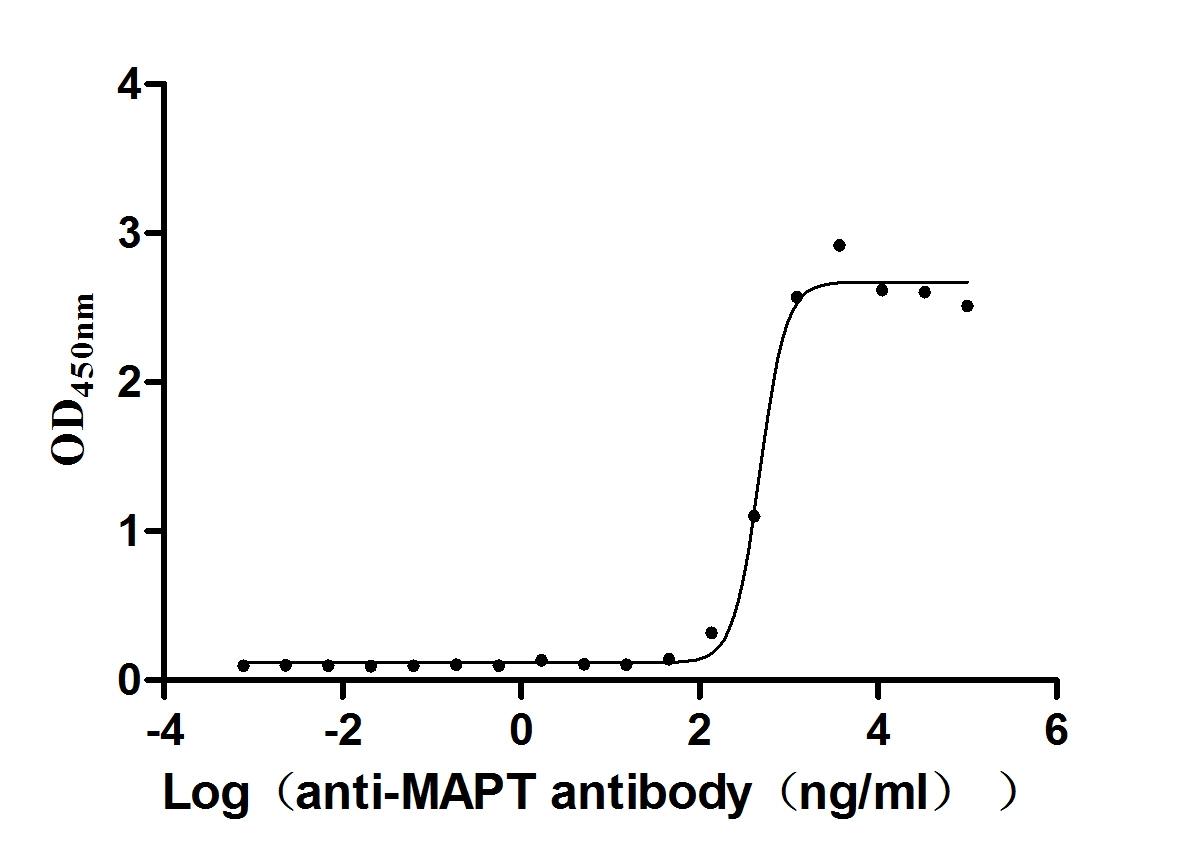
Functional validation through ELISA confirms its specific binding capability to anti-Mapt antibody (CSB-RA013481A1HU) with an EC50 range of 436.1-518.6 ng/mL when immobilized at 2 μg/mL. The mammalian expression system ensures proper post-translational modifications and folding characteristics critical for maintaining Mapt's native biological functions.
Presented as lyophilized powder, this protein offers excellent stability and convenient handling. The N-terminal 10×His tag facilitates purification without interfering with functional domains. This preparation is particularly valuable for Alzheimer's disease and tauopathy research, microtubule dynamics studies, neurodegenerative disease modeling, and therapeutic development for tau-related disorders.
The mouse Mapt protein, encoded by the Mapt gene, is a crucial component of the microtubule-associated protein family and specifically encodes the Tau protein. This protein plays a significant role in stabilizing microtubules, which are essential for maintaining cellular structure and intracellular transport within neurons [1]. In various studies, Mapt has been implicated in several neurodegenerative diseases, most notably tauopathies, which include conditions such as Alzheimer's disease (AD) and frontotemporal dementia (FTD) [2].
Transgenic mouse models that express various human Mapt mutations, such as the P301L mutation, have been instrumental in elucidating the pathogenic mechanisms underlying tauopathies [3]. For instance, P301L transgenic mice exhibit characteristic neurofibrillary tangles and cognitive deficits that parallel findings in human patients with tauopathies [4]. Additionally, the manipulation of Mapt expression in these models has revealed insights into synaptic dysfunction and impaired glutamate dynamics, linking Mapt expression levels to neurodegenerative processes [3][5].
Furthermore, studies indicate that the absence or downregulation of Mapt can influence neuroinflammatory responses as well as neuronal excitability. For instance, Mapt deletion in specific mouse models has been shown to mitigate certain forms of epilepsy and hyperexcitability, suggesting a complex role for Tau in modulating neuronal activity beyond mere structural support [6]. This aspect of Mapt research highlights its potential as a therapeutic target; studies have indicated that reducing Mapt expression may protect against cognitive deficits associated with Alzheimer's disease in mouse models [2][7].
The diversity of isoforms produced by the Mapt gene due to alternative splicing results in different tau protein variants that can exhibit distinct biological properties and functions [8]. These differences are critical when considering the pathological roles of tau in various tauopathies, as certain isoforms may be more prone to aggregation and, therefore, more detrimental to neuronal health [9].
References:
[1] I. Avitan, Y. Halperin, et al. Towards a consensus on alzheimer’s disease comorbidity? Journal of Clinical Medicine, vol. 10, no. 19, p. 4360, 2021. https://doi.org/10.3390/jcm10194360
[2] X. Da, S. Besselink, et al. Programmable epigenome editing by transient delivery of crispr epigenome editor ribonucleoproteins. 2024. https://doi.org/10.1101/2024.11.26.625496
[3] R. Crescenzi, C. DeBrosse, et al. Longitudinal imaging reveals subhippocampal dynamics in glutamate levels associated with histopathologic events in a mouse model of tauopathy and healthy mice. Hippocampus, vol. 27, no. 3, p. 285-302, 2017. https://doi.org/10.1002/hipo.22693
[4] M. Fernández‐Nogales, M. Santos‐Galindo, et al. Tau‐positive nuclear indentations in p301s tauopathy mice. Brain Pathology, vol. 27, no. 3, p. 314-322, 2016. https://doi.org/10.1111/bpa.12407
[5] C. Chen, J. Holth, et al. mapt deletion fails to rescue premature lethality in two models of sodium channel epilepsy. Annals of Clinical and Translational Neurology, vol. 5, no. 8, p. 982-987, 2018. https://doi.org/10.1002/acn3.599
[6] J. Hull, H. O’Malley, et al. Excitatory and inhibitory neuron defects in a mouse model of scn1b‐linked eiee52. Annals of Clinical and Translational Neurology, vol. 7, no. 11, p. 2137-2149, 2020. https://doi.org/10.1002/acn3.51205
[7] T. Taniguchi, N. Doe, et al. Transgenic mice expressing mutant (n279k) human tau show mutation dependent cognitive deficits without neurofibrillary tangle formation. Febs Letters, vol. 579, no. 25, p. 5704-5712, 2005. https://doi.org/10.1016/j.febslet.2005.09.047
[8] Y. Pan, Y. Pan, Y. Cheng, F. Yang, Z. Yao, & O. Wang Knockdown of lncrna mapt-as1 inhibites proliferation and migration and sensitizes cancer cells to paclitaxel by regulating mapt expression in er-negative breast cancers. Cell & Bioscience, vol. 8, no. 1, 2018. https://doi.org/10.1186/s13578-018-0207-5
[9] T. Müller, A. Schrötter, et al. Sense and nonsense of pathway analysis software in proteomics. Journal of Proteome Research, vol. 10, no. 12, p. 5398-5408, 2011. https://doi.org/10.1021/pr200654k